
Chapter 9 345
Copyright © 2017 Pearson Education, Inc.
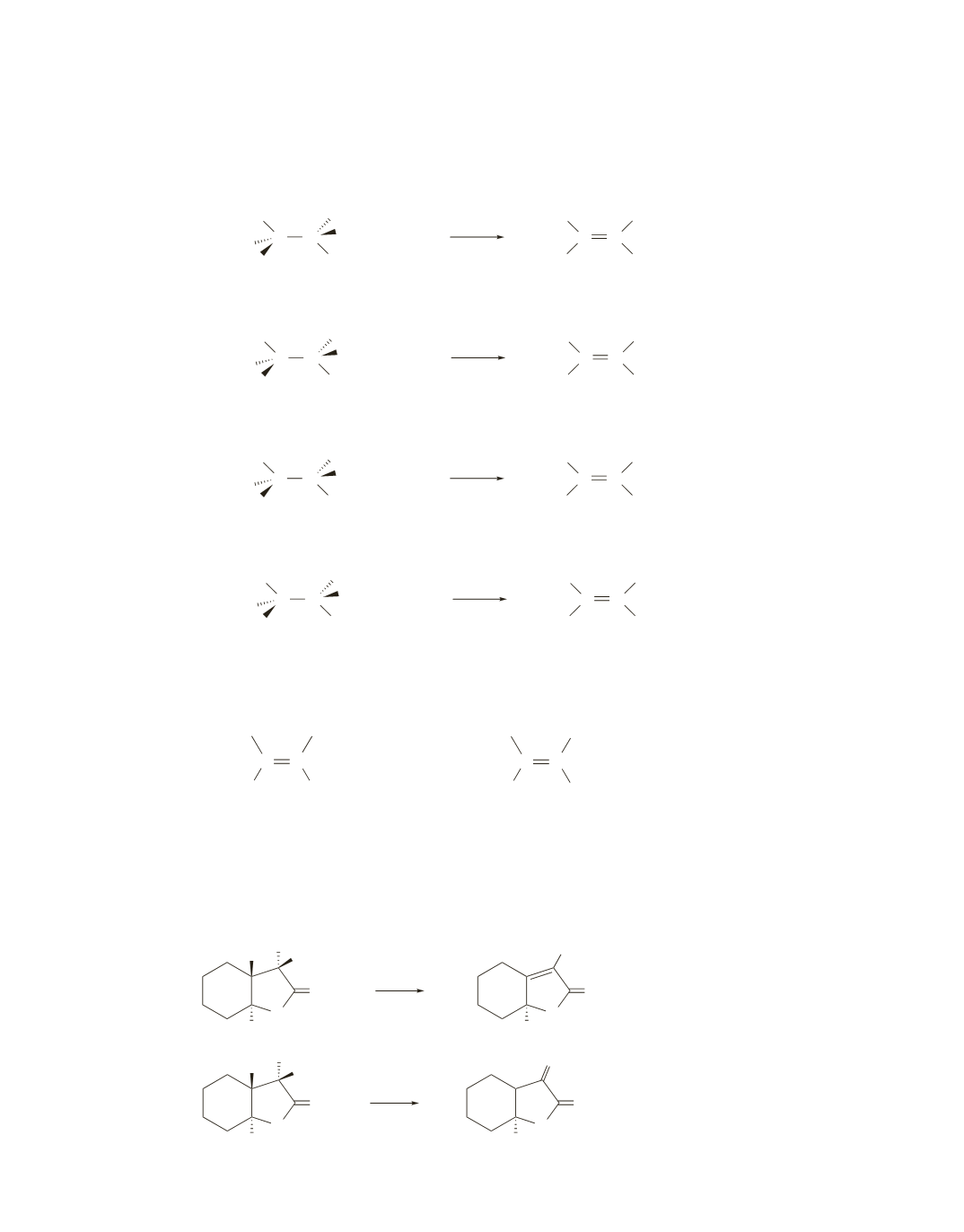
111.
In reactions
a–d
, the
b
-carbon from which the hydrogen is to be removed is bonded to only one hydrogen.
Therefore, the configuration of the reactant determines the configuration of the product of the E2 reaction.
To determine the configuration of the product, see the instructions in the Problem-Solving Strategy on page 425.
a.
H
CH
3
CH
3
CH
2
CH
3
(
E
)-3-methyl-2-pentene
(2
S
,3
S
)-2-chloro-3-methylpentane
C C
H
CH
3
CH
3
CH
2
CH
3
C C
Cl
H
b.
H
CH
3
CH
3
CH
2
CH
3
(
Z
)-3-methyl-2-pentene
(2
S
,3
R
)-2-chloro-3-methylpentane
C C
H
CH
3
CH
3
CH
2
CH
3
C C
Cl
H
c.
H
CH
3
CH
3
CH
2
CH
3
(
Z
)-3-methyl-2-pentene
(2
R
,3
S
)-2-chloro-3-methylpentane
C C
H
CH
3
CH
3
CH
2
CH
3
C C
Cl
H
d.
H
CH
3
CH
3
CH
2
CH
3
(
E
)-3-methyl-2-pentene
(2
R
,3
R
)-2-chloro-3-methylpentane
C C
H
CH
3
CH
3
CH
2
CH
3
C C
Cl
H
e.
This reactant has two hydrogens bonded to the
b
-carbon, so both
Z
and
E
stereoisomers are formed.
+
CH
3
CH
2
(CH
3
)
3
C
H
CH
3
CH
3
CH
2
C C
C C
(CH
3
)
3
C
CH
3
H
major product
minor product
112.
The two tertiary alkyl halides form different products when they react with a strong base (an E2 reaction)
because the H and Br attached to the ring can be eliminated only if they are anti to each other. Because
the H and Br are on the same side of the ring in
b
, a hydrogen has to be removed from the other
b
-carbon (that is, from the methyl group).
a.
O
H
H
Br
CH
3
O
O
H
O
CH
3
b.
O
H
H
CH
3
Br
O
O
H
O
CH
2
















