
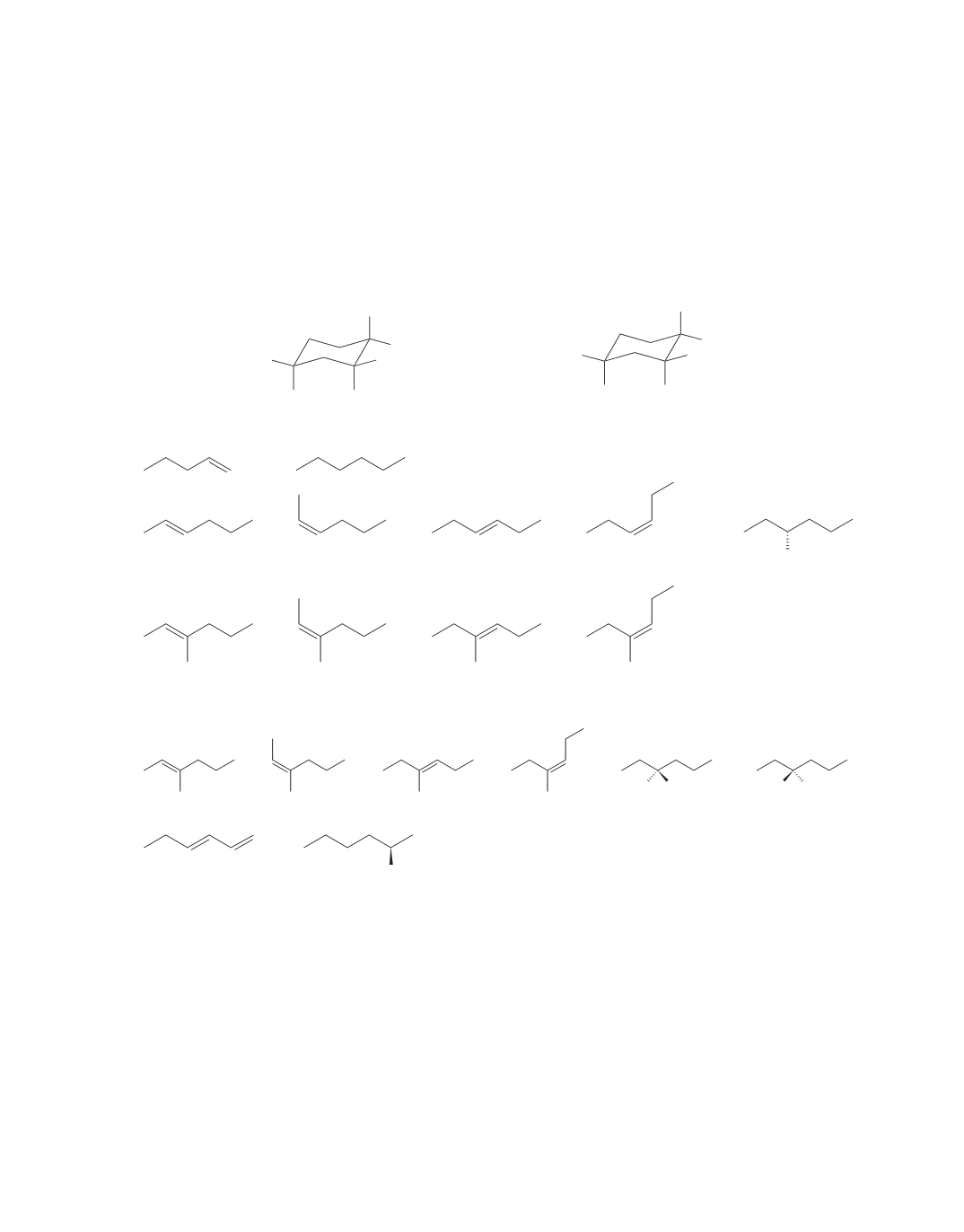
342 Chapter 9
Copyright © 2017 Pearson Education, Inc.
102.
In an E2 reaction, both groups to be eliminated must be in axial positions.
When the bromine is in the axial position in the cis isomer, the
tert
-butyl substituent is in the more stable
equatorial position.
When the bromine is in the axial position in the trans isomer, the
tert
-butyl substituent is in the less stable
axial position.
Therefore, elimination takes place via the most stable conformer in the cis isomer and via the less stable
chair conformer in the trans isomer, so the cis isomer undergoes elimination more rapidly.
H H
(CH
3
)
3
C
H
Br
H
(CH
3
)
3
C H
H
H
Br
H
cis
-1-bromo-4-
tert
-butylcyclohexane
trans
-1-bromo-4-
tert
-butylcyclohexane
103.
a.
+
OCH
3
b.
major
minor
+
major
minor
+
+
+
OCH
3
c.
minor
major
+
+
major
minor
+
d.
+
minor
major
+
major
minor
+
+
OCH
3
+
CH
3
O
e.
+
OCH
3
f.
no substitution or elimination reaction
104.
The reactants are neutral. Therefore, increasing the polarity of the solvent increases the rate of the reactions.
Therefore, the reaction is faster in formic acid, the more polar of the two solvents.
















