
338 Chapter 9
Copyright © 2017 Pearson Education, Inc.
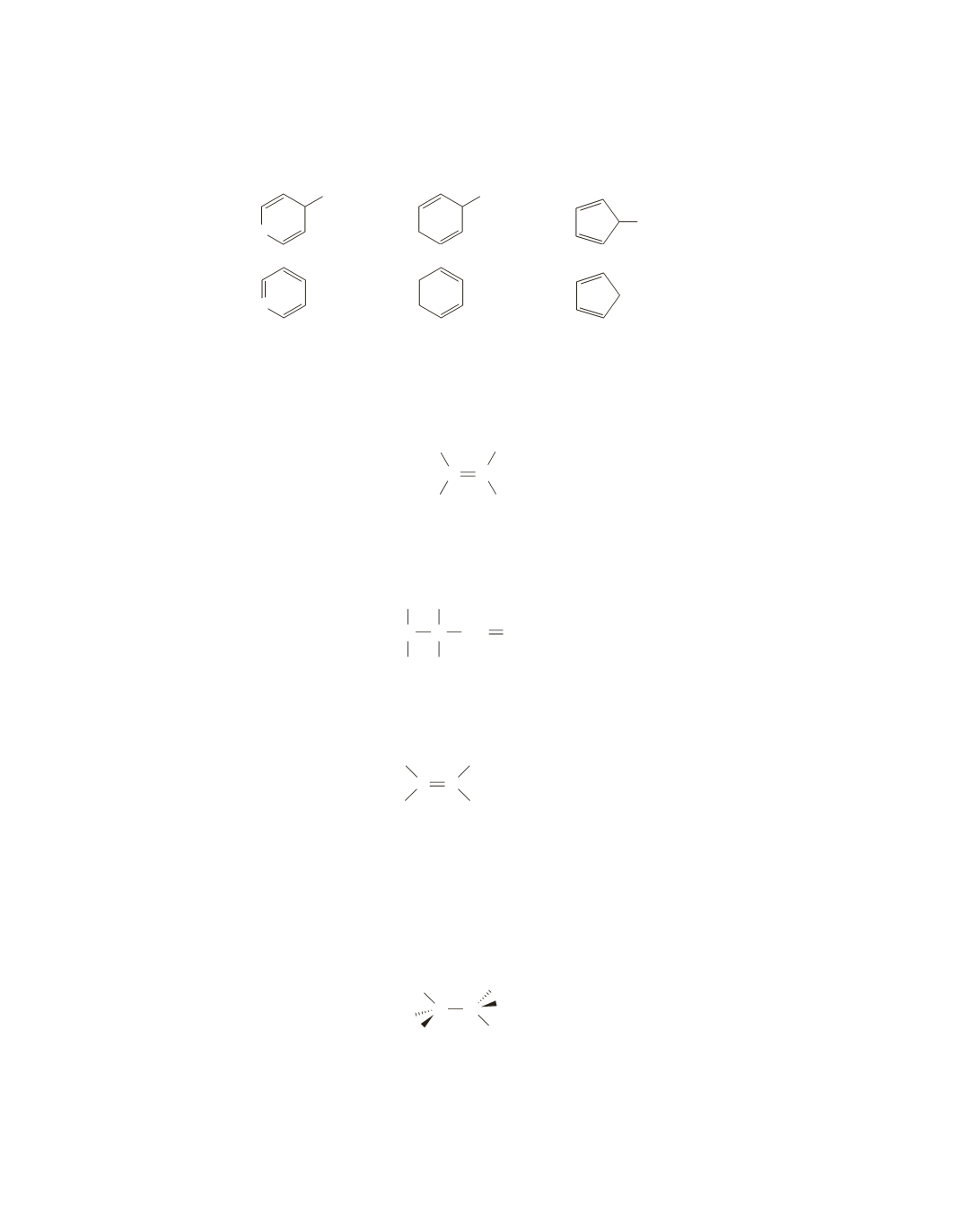
95.
Draw the carbocation that each compound forms in an
S
N
1
reaction. Because carbocation formation is the
rate-limiting step, the more stable the carbocation, the faster is the rate of the
S
N
1
reaction.
>
>
O
Cl
Cl
Cl
O
most stable carbocation
because it is aromatic
least stable carbocation
because it is antiaromatic
+
+
+
96.
a.
The stereoisomer formed in greatest yield is the one in which the larger group attached to one
sp
2
carbon and the larger group attached to the other
sp
2
carbon are on opposite sides of the double bond.
C C
(CH
3
)
3
C
H
CH
3
CH
3
b.
No stereoisomers are possible for this compound because one of the
sp
2
carbons is bonded to two
hydrogens.
CH CH
2
C CH
3
C
CH
3
CH
3
CH
3
CH
3
c.
No stereoisomers are possible for this compound because one of the
sp
2
carbons is bonded to two
methyl groups.
C C
CH
2
CH
3
CH
3
CH
3
CH
3
d.
Because it is an E2 reaction and only one hydrogen is attached to the
b
-carbon, the stereoisomer
formed in greater yield depends on the configuration of the reactant. The reactant can have four differ-
ent configurations:
S,S
;
S,R
;
R,R
; and
R,S
. To determine the product of the reaction:
1.
Draw the skeleton of a perspective formula, putting the groups to be eliminated on the solid lines.
Notice that on each carbon, the solid wedge is below the hatched wedge.
C C
Br
H
















