
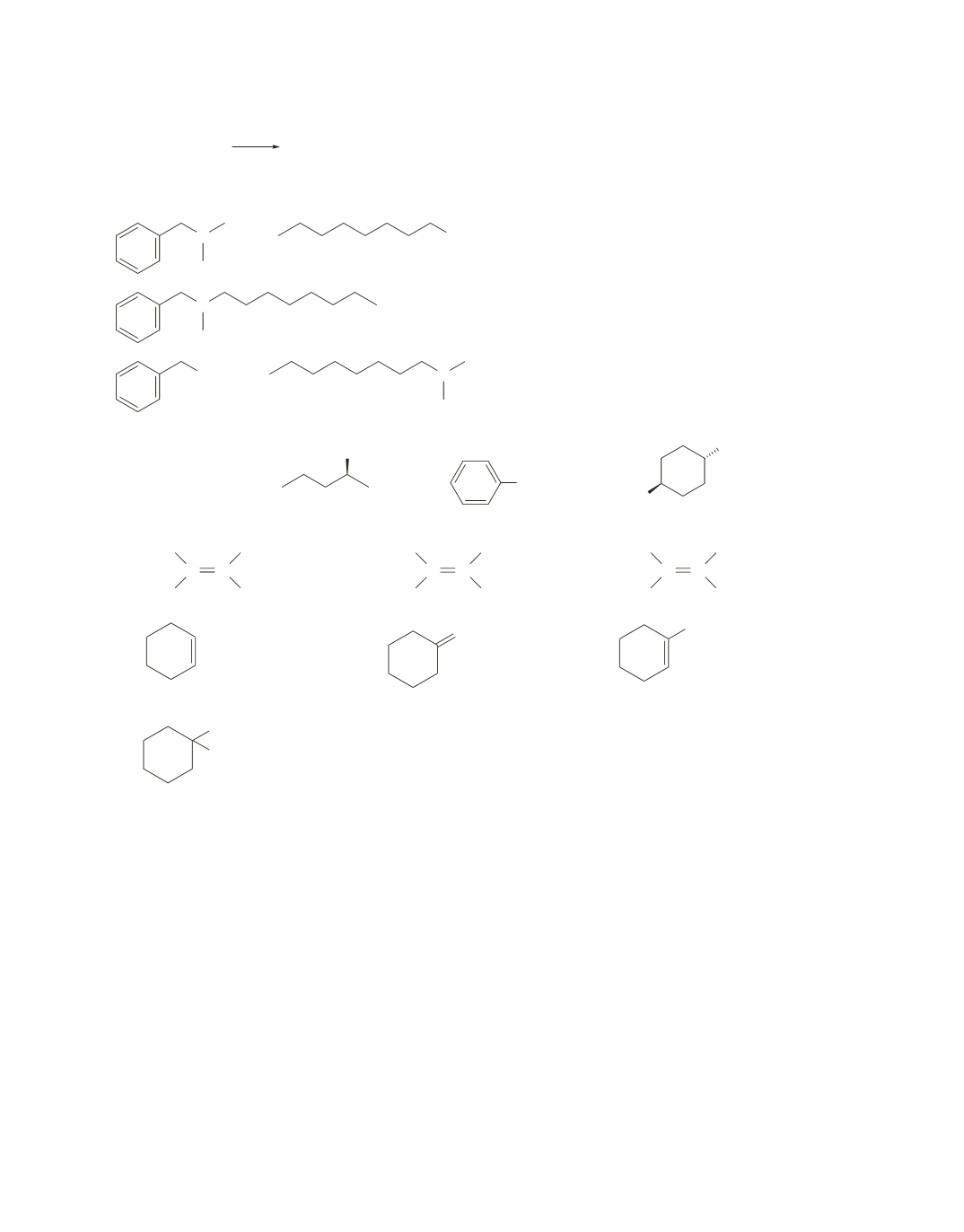
334 Chapter 9
Copyright © 2017 Pearson Education, Inc.
d.
(CH
3
)
3
COH
+
HBr
H
2
O
(CH
3
)
3
CBr
Because the reactants are neutral,
the reaction is faster in the more
polar solvent.
81.
+
N
+
+
Cl
N
CH
3
Cl
N
Cl
82.
a.
CH
3
O
-
b.
Br
c.
O
−
d.
CH
3
Br
83.
a.
C C
H
CH
3
H
3
C
H
b.
c.
C C
H
CH
3
H
3
C
H
d.
CH
2
e.
C C
H
CH
2
CH
3
H
3
C
H
f.
CH
3
84.
f.
CH
Cl
3
This is the only one that can undergo an E1 reaction.
85.
a. 1.
An E1 reaction is not affected by the strength of the base but, if the reaction can take place by both
an E1 and E2 reaction, a weak base will favor the E1 reaction by disfavoring an E2 reaction.
2.
An E1 reaction is not affected by the concentration of the base but, if the reaction can take place by
both an E1 and E2 reaction, a low concentration of a base will favor the E1 reaction by disfavoring
an E2 reaction.
3.
If the reactant is charged, an E1 reaction will be favored by the least polar solvent that will dissolve
the reactant (generally an aprotic polar solvent). If the reactant is not charged, an E1 reaction will
be favored by a protic polar solvent.
b. 1.
A strong base favors an E2 reaction.
2.
A high concentration of a base favors an E2 reaction.
3.
If one of the reactants is charged, an E2 reaction will be favored by the least polar solvent that will
dissolve the reactant (generally an aprotic polar solvent). If neither of the reactants is charged, an
E2 reaction will be favored by a protic polar solvent.
















