
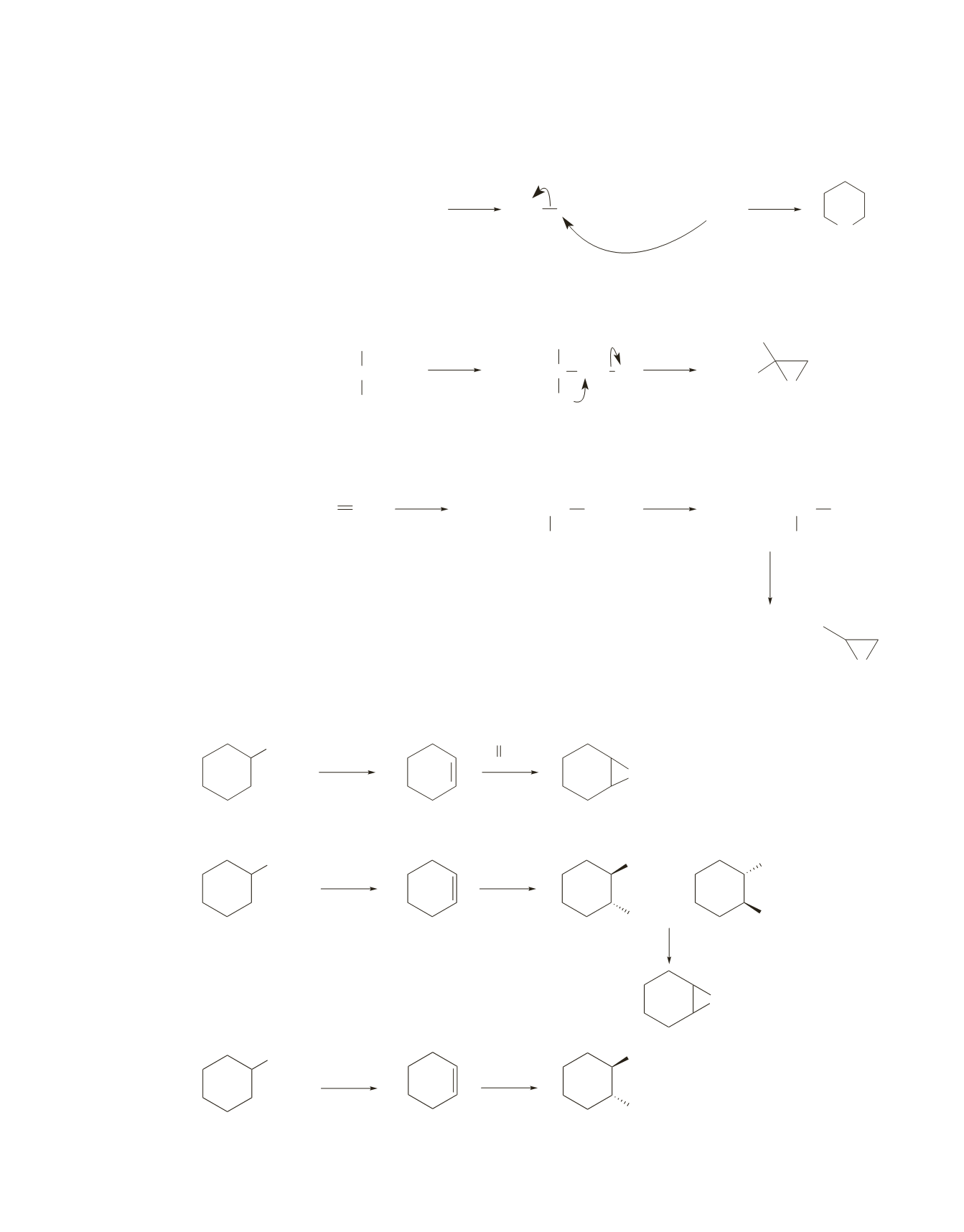
Chapter 9 329
Copyright © 2017 Pearson Education, Inc.
c.
Hydride ion removes a proton from the OH group and the alkoxide ion attacks the back side of the
carbon attached to the bromine, forming a six-membered ring ether.
BrCH
2
CH
2
CH
2
CH
2
CH
2
OH
CH
2
CH
2
CH
2
CH
2
CH
2
O
Br
.. .. ..
_
O
NaH
d.
Hydride ion removes a proton from the OH group and the alkoxide ion attacks the back side of the
carbon attached to the chlorine, forming an epoxide.
CH
3
CH
2
CCH
2
Cl
CH
3
OH
CH
3
CH
2
C
CH
3
O
CH
2
Cl
..
_
.. ..
O
CH
3
CH
2
CH
3
NaH
e.
After the halohydrin is formed, hydride ion removes a proton from the OH group and the alkoxide ion
forms an epoxide.
O
CH
3
CH
2
CH
2
CH CH
2
Cl
2
H
2
O
CH
3
CH
2
CH
2
CH CH
2
Cl
OH
CH
3
CH
2
CH
2
NaH
CH
3
CH
2
CH
2
CH CH
2
Cl
O
−
66.
In parts
a
and
b
, a bulky base is used to encourage elimination over substitution.
a.
O
tert
-BuO
−
RCOOH
O
or
Br
Br
Br
OH
OH
Br
O
Br
2
H
2
O
NaH
+
tert
-BuO
−
b.
Br
Br
Br
tert
-BuO
−
Br
2
















