
Chapter 9 327
Copyright © 2017 Pearson Education, Inc.
57.
Solved in the text.
58.
Acetate ion is a better nucleophile in dimethyl sulfoxide because dimethyl sulfoxide does not stabilize the
negatively charged nucleophile by ion–dipole interactions, whereas methanol does stabilize it by ion–dipole
interactions.
59.
Only an
S
N
1
reaction gives the product with retention of configuration. Therefore, water (a poor nucleo-
phile) should be used for the reaction.
60.
Because a strong base is used in the Williamson ether synthesis, the reaction is an
S
N
2
reaction, so a
competing E2 reaction can also occur. The elimination product is a minor product because substitution is
favored when the alkyl halide is primary.
a.
−
+
+
b.
−
+
+
61.
To maximize the amount of ether formed in the
S
N
2
reaction, make sure the less hindered group is
provided by the alkyl halide. In order to convert the alcohol
1
p
K
a
15
2
to an alkoxide ion in a reaction
that favors products (in parts
a
,
b
, and
c
), a strong base
1
H
-
2
is needed.
a.
CH
3
CH
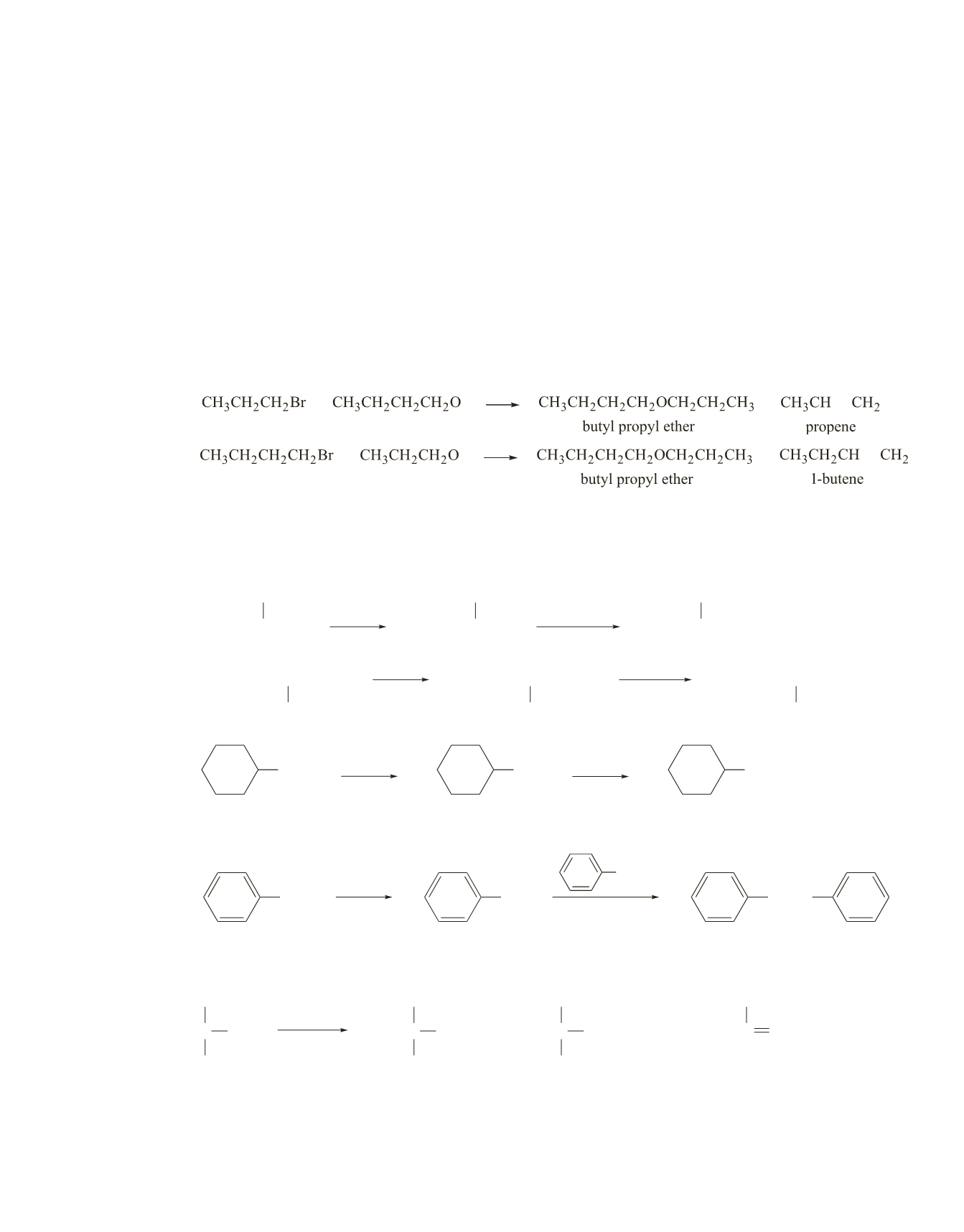
2
CHOH
CH
3
NaH
CH
3
CH
2
CHO
−
CH
3
CH
3
CH
2
CH
2
Br
CH
3
CH
2
CHOCH
2
CH
2
CH
3
CH
3
b.
CH
3
CH
2
CH
2
CHCH
2
OH
CH
3
NaH
CH
3
CH
2
CH
2
CHCH
2
O
−
CH
3
CH
3
CH
2
Br
CH
3
CH
2
CH
2
CHCH
2
OCH
2
CH
3
CH
3
c.
OH
O
−
OCH
3
NaH
CH
3
Br
In part
d
,
HO
-
can be used to convert phenol
1
p
K
a
15
2
to phenoxide ion in a reaction that favors
products, because phenol is a considerably stronger acid than an alcohol.
d.
OH
NaOH
O
−
CH
2
Br
CH
2
O
Remember that aryl halides cannot undergo
S
N
2
reactions.
62.
CH
3
CH
3
C Br
CH
3
CH
3
CH
2
OH
H
2
O
CH
3
CH
3
C OH
CH
3
CH
3
CH
3
C OCH
2
CH
3
CH
3
+
+
CH
3
C CH
2
CH
3
















