
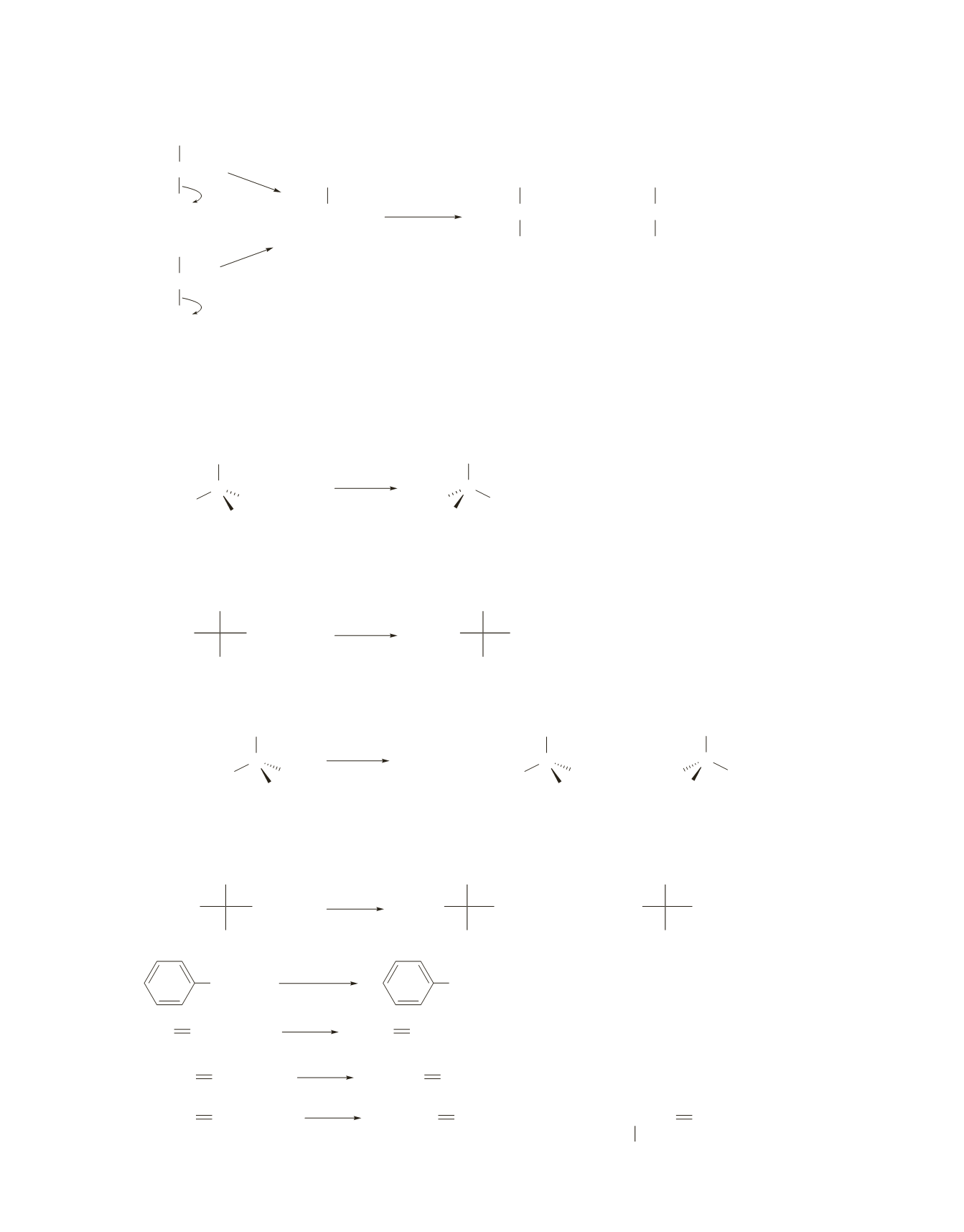
332 Chapter 9
Copyright © 2017 Pearson Education, Inc.
76.
a.
CH
3
CCH
3
CH
3
Br
CH
3
CCH
3
CH
3
+
CH
3
CH
2
OH
H
2
O
CH
3
CCH
3
CH
3
OCH
2
CH
3
+
CH
3
CCH
3
CH
3
OH
CH
3
CCH
3
CH
3
Cl
Br
−
Cl
−
b.
The products are obtained as a result of the nucleophiles reacting with the carbocation. 2-Bromo-
2-methylpropane and 2-chloro-2-methylpropane form the same carbocation, so both alkyl halides form
the same products.
77.
a.
The
S
N
2
reaction takes place with inversion of configuration.
C
CH
2
CH
2
CH
3
CH
3
H OCH
3
C
CH
2
CH
2
CH
3
CH
3
H Br
CH
3
O
−
(
R
)-2-bromopentane
(
S
)-2-methoxypentane
CH
3
H
Br
CH
2
CH
2
CH
3
CH
3
OCH
3
H
CH
2
CH
2
CH
3
or
CH
3
O
−
b.
The S
N
1 reaction takes place with inversion of configuration.
or
CH
3
OH
(
R
)-3-bromo-3-methylheptane
(
S
)-3-methoxy-3-methylheptane
C
CH
2
CH
3
Br
CH
3
CH
3
CH
2
CH
2
CH
2
(
R
)-3-methoxy-3-methylheptane
C
CH
2
CH
3
OCH
3
CH
3
CH
2
CH
2
CH
2
C
CH
2
CH
3
CH
3
O
CH
3
CH
2
CH
2
CH
2
CH
3
+
CH
2
CH
3
CH
3
Br
CH
2
CH
2
CH
2
CH
3
CH
2
CH
3
CH
3
CH
3
O
CH
2
CH
2
CH
2
CH
3
CH
2
CH
3
OCH
3
CH
3
CH
2
CH
2
CH
2
CH
3
CH
3
OH
+
CH
3
c.
CH
3
CH
2
OH
CH
2
Cl
CH
2
OCH
2
CH
3
d.
CH
2
CHCH
2
Cl
CH
2
CHCH
2
OCH
3
CH
3
OH
e.
CH
3
CH CHCH
2
Br
CH
3
CH CHCH
2
OCH
3
CH
3
O
−
f.
CH
3
CH CHCH
2
Br
CH
3
CH CHCH
2
OCH
3
CH
3
CHCH
OCH
3
CH
2
CH
3
OH
+
















