
Chapter 9 337
Copyright © 2017 Pearson Education, Inc.
90.
a.
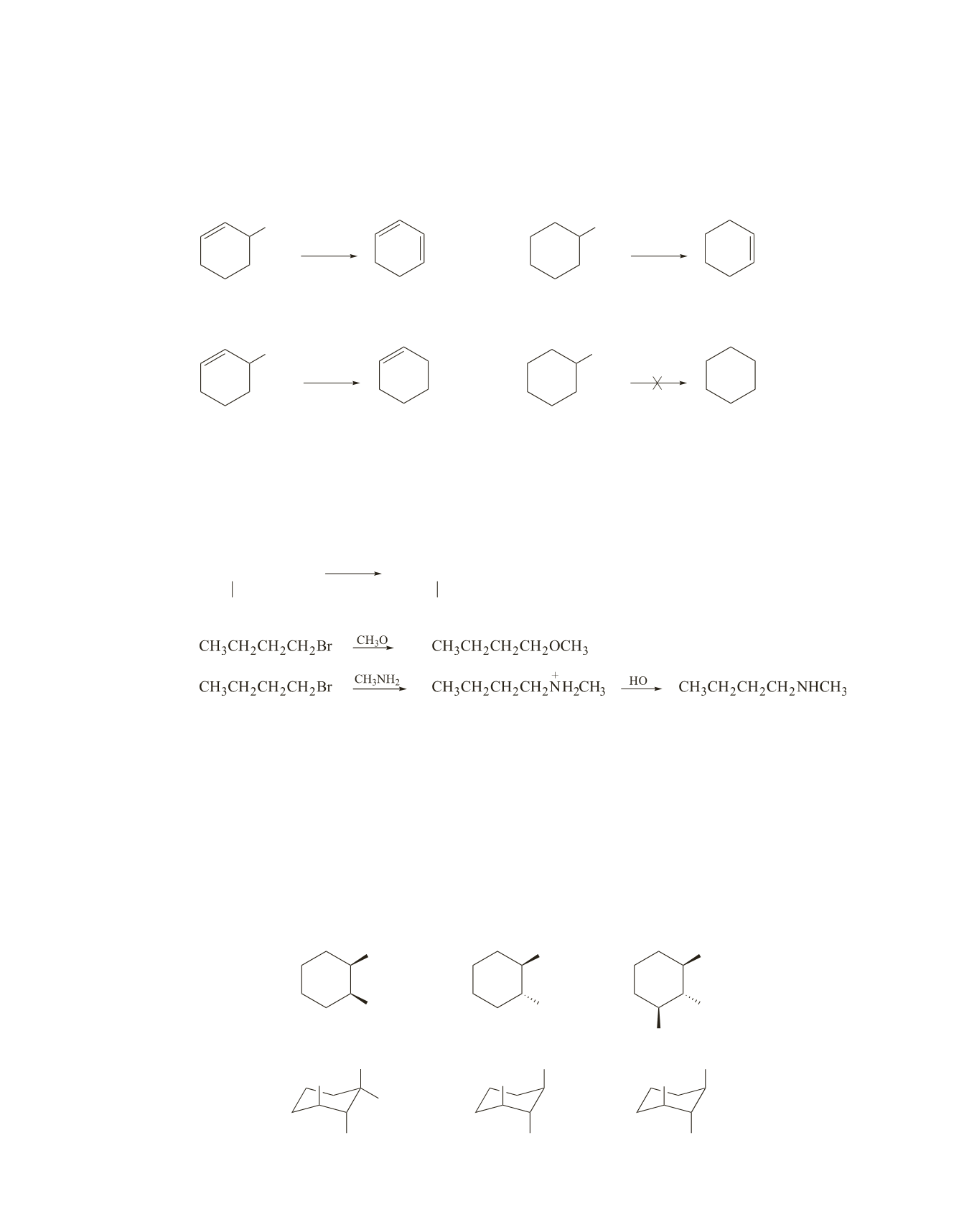
3-Bromocyclohexene forms 1,3-cyclohexadiene; bromocyclohexane forms cyclohexene. 3-Bromo-
cyclohexene reacts faster in an E2 reaction, because a conjugated double bond is more stable than an
isolated double bond. So the transition state leading to formation of the conjugated double bond is
more stable and, therefore, the conjugated double bond is easier to form.
Br
E2
Br
E2
base
base
b.
3-Bromocyclohexene, because it forms a relatively stable secondary allylic cation. The other com-
pound is a secondary alkyl halide, so it does not undergo an E1 reaction.
Br
E1
E1
Br
+
+
91.
That fact that the change from hydrogen to deuterium affects the rate of the reaction tells us that the C–H
(or C–D) bond is broken in the rate-limiting step. This is consistent with the mechanism for an E2 reaction
but not for the mechanism for an E1 reaction.
92.
Alkyl chlorides and alkyl iodides could also be used. Do not use alkyl fluorides because they have the
poorest leaving groups and cannot undergo
S
N
2
reactions.
a.
CH
3
CHCH
2
CH
3
CH
3
CHCH
2
CH
3
OCH
3
CH
3
Br
−
O
b.
−
c.
−
93.
a.
ethoxide ion, because elimination is favored by bulky bases and
tert
-butoxide ion is bulkier than
ethoxide ion
b.
-
SCN
because elimination is favored by strong bases and
-
OCN
is a stronger base than
-
SCN
c.
Br
-
because elimination is favored by strong bases and
Cl
-
is a stronger base than
Br
-
d.
CH
3
S
-
because elimination is favored strong bases and
CH
3
O
-
is a stronger base than
CH
3
S
-
94.
The first compound listed below is the most reactive compound because it has two axial hydrogens attached
to
b
-carbons. The second compound has one axial hydrogen attached to a
b
-carbon, but it cannot form the
more substituted (more stable) alkene that can be formed by the first compound. The last compound cannot
undergo an E2 reaction because it does not have an axial hydrogen attached to a
b
-carbon.
Br
CH
3
>
Br
CH
3
>
Br
CH
3
CH
3
H
H
Br
CH
3
>
H
CH
3
Br
>
CH
3
CH
3
Br
















