
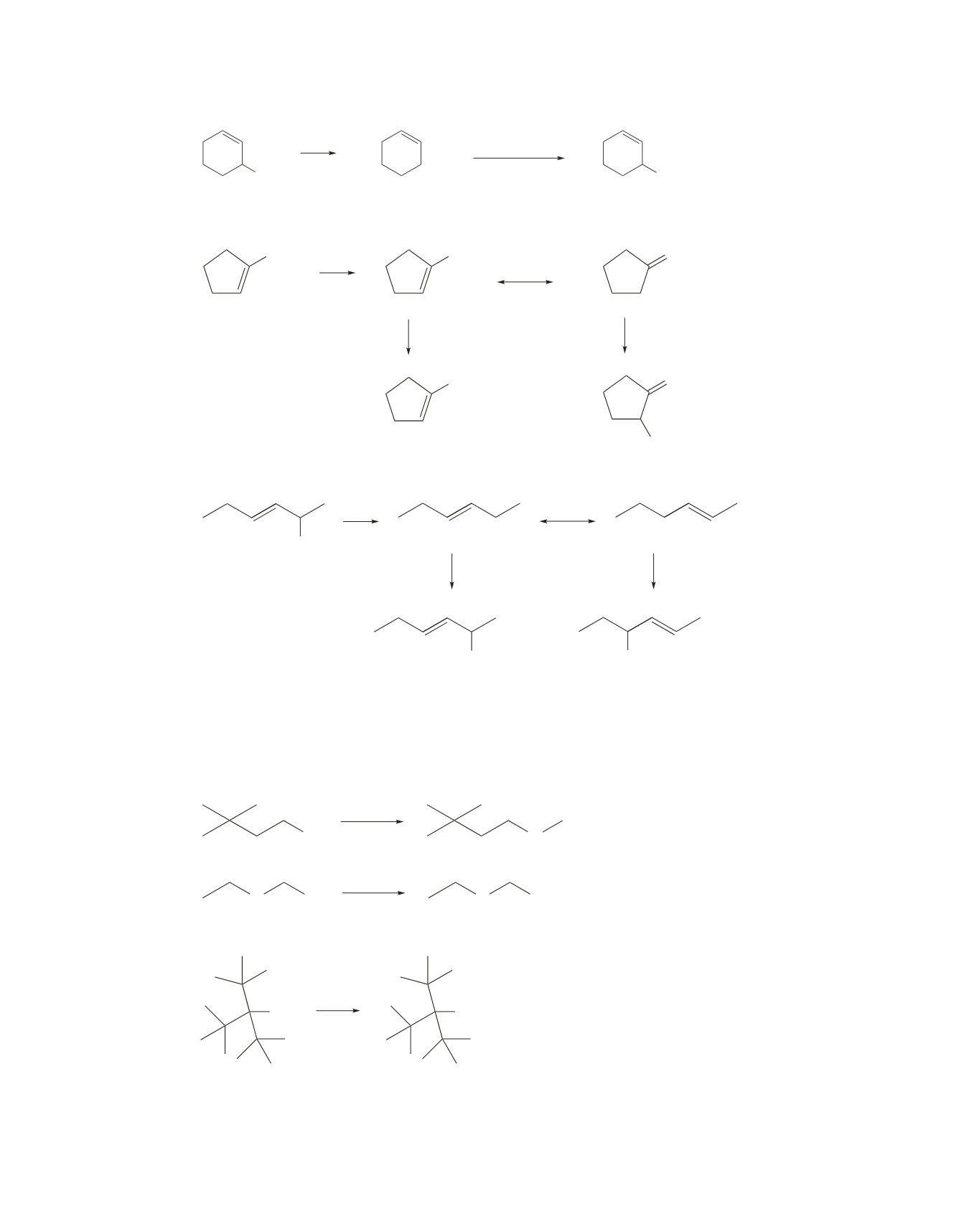
Chapter 9 333
Copyright © 2017 Pearson Education, Inc.
78.
a.
79.
Methoxide ion is a better nucleophile in DMSO, because DMSO cannot stabilize the ion by ion–dipole
interactions.
80.
a.
CH
3
S
−
+
Cl
−
Cl
S
The nucleophile is less sterically
hindered.
b.
HO
−
+
Cl
−
O
O OH
Cl
H
2
O
The electron-withdrawing oxygen
increases the electrophilicity of the
carbon that the nucleophile attacks.
c.
Cl
OH
+
H
3
O
+
Cl
+
−
H
2
O
Steric strain is decreased when the
alkyl halide dissociates to form the
carbocation because the hybridiza-
tion of the carbon changes from
sp
3
to
sp
2
,
allowing the bond angle
between the bulky groups to increase
from
109.5
°
to
120
°
, which allows
more room for the bulky groups.
Relieving steric strain causes the car-
bocation to be formed more rapidly.
b.
c.
Br
CH
2
Br
CH
2
+
CH
2
+
CH
3
CH
2
OH
+
+
Br
−
CH
3
CH
2
OH
CH
2
OCH
2
CH
3
OCH
2
CH
3
CH
3
CH OH
2
CH
2
OCH
2
CH
3
+
Br
CH
3
CH
2
OH
Br
−
CH
3
CH
2
OH
+
+
Both the
R
and
S
stereoisomers are obtained.
Both the
R
and
S
stereoisomers are obtained.
OCH
2
CH
3
OCH
2
CH
3
This compound has an asymmetric
center, so both the
R
and
S
stereoisomers are obtained.
This compound has an asymmetric
center, so both the
R
and
S
stereoisomers are obtained.
Br
CH
2
Br
CH
2
+
CH
2
+
CH
3
CH
2
OH
+
+
Br
−
CH
3
CH
2
OH
CH
2
OCH
2
CH
3
OCH
2
CH
3
CH
3
CH OH
2
CH
2
OCH
2
CH
3
+
Br
CH
3
CH
2
OH
Br
−
CH
3
CH
2
OH
+
+
Both the
R
and
S
stereoisomers are obtained.
Both the
R
and
S
stereoisomers are obtained.
OCH
2
CH
3
OCH
2
CH
3
This compound has an asymmetric
center, so both the
R
and
S
stereoisomers are obtained.
This compound has an asymmetric
center, so both the
R
and
S
stereoisomers are obtained.
















