
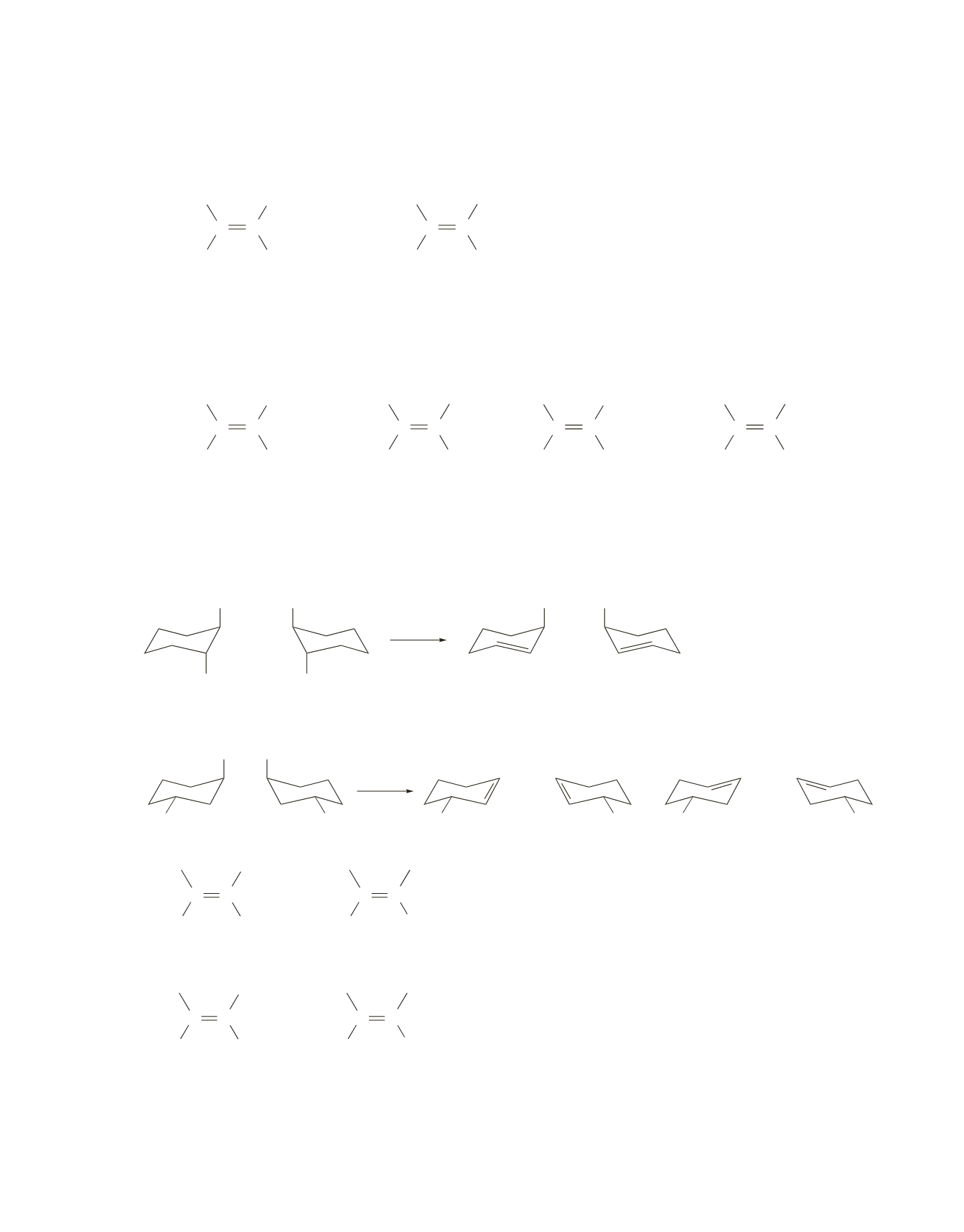
336 Chapter 9
Copyright © 2017 Pearson Education, Inc.
89.
The very minor products that are obtained from “anti-Zaitsev” elimination (that is, the less substituted
alkenes) are not shown.
a.
CH
3
CH
2
CH
2
C C
H
CH
3
H
+
minor
CH
3
CH
2
CH
2
C C
H
H
CH
3
major
b.
The reactant has two
b
-carbons that are attached to two hydrogens, so the reaction is not regioselec-
tive. Therefore, two constitutional isomers can be formed. Each constitutional isomer has
E
and
Z
stereoisomers.
CH
3
CH
2
CH
2
C C
CH
3
H
+
+
minor
CH
3
CH
2
CH
2
C C
H
CH
3
major
CH
3
CH
2
C C
CH
3
CH
2
CH
2
CH
3
H
+
minor
C C
H
CH
2
CH
3
major
CH
3
CH
3
CH
3
CH
3
c.
trans
-1-Chloro-2-methylcyclohexane has two stereoisomers.
A hydrogen cannot be removed from the
b
-carbon bonded to the fewest hydrogens because that hydrogen
is not attached to an axial bond.
Cl
CH
3
Cl
CH
3
CH
3
CH
3
+
CH
3
O
−
+
d.
trans
-1-Chloro-3-methylcyclohexane has two stereoisomers, and each can form two elimination products.
Cl
CH
3
CH
3
Cl
+
+
+
CH
3
O
−
CH
3
CH
3
+
CH
3
CH
3
e.
CH
3
CH
2
C C
CH
3
CH
3
CH
3
H
CH
3
CH
2
+
C C
H
CH
3
minor
major
f.
CH
3
CH
3
CH
3
CH
2
CH
3
H
CH
3
CH
2
+
C C
C C
H
CH
3
minor
major
















