
324 Chapter 9
Copyright © 2017 Pearson Education, Inc.
39.
a.
CH
3
CH
2
CH
2
Br
This compound has less steric hindrance.
b.
CH
3
I
I
-
is a better leaving group (weaker base) than
Br
-
.
c.
CH
3
CH
2
CH
2
CCH
3
CH
3
Br
The tertiary alkyl halide because a secondary alkyl halide does not
undergo
S
N
1
reactions.
40.
The reaction of an alkyl halide with an acetylide ion is an
S
N
2
reaction. Methyl and primary alkyl halides
work best because they have the least steric hindrance to back-side attack. In addition, primary alky halides
form mainly the desired substitution product and methyl halides form only the desired substitution product.
41.
Because
CH
3
S
-
is a better nucleophile in the protic polar solvent and a weaker base than
CH
3
O
-
,
the ratio
of substitution (where
Y
-
reacts as a nucleophile) to elimination (where
Y
-
reacts as a base) increases
when the nucleophile is changed from
CH
3
O
-
to
CH
3
S
-
.
42.
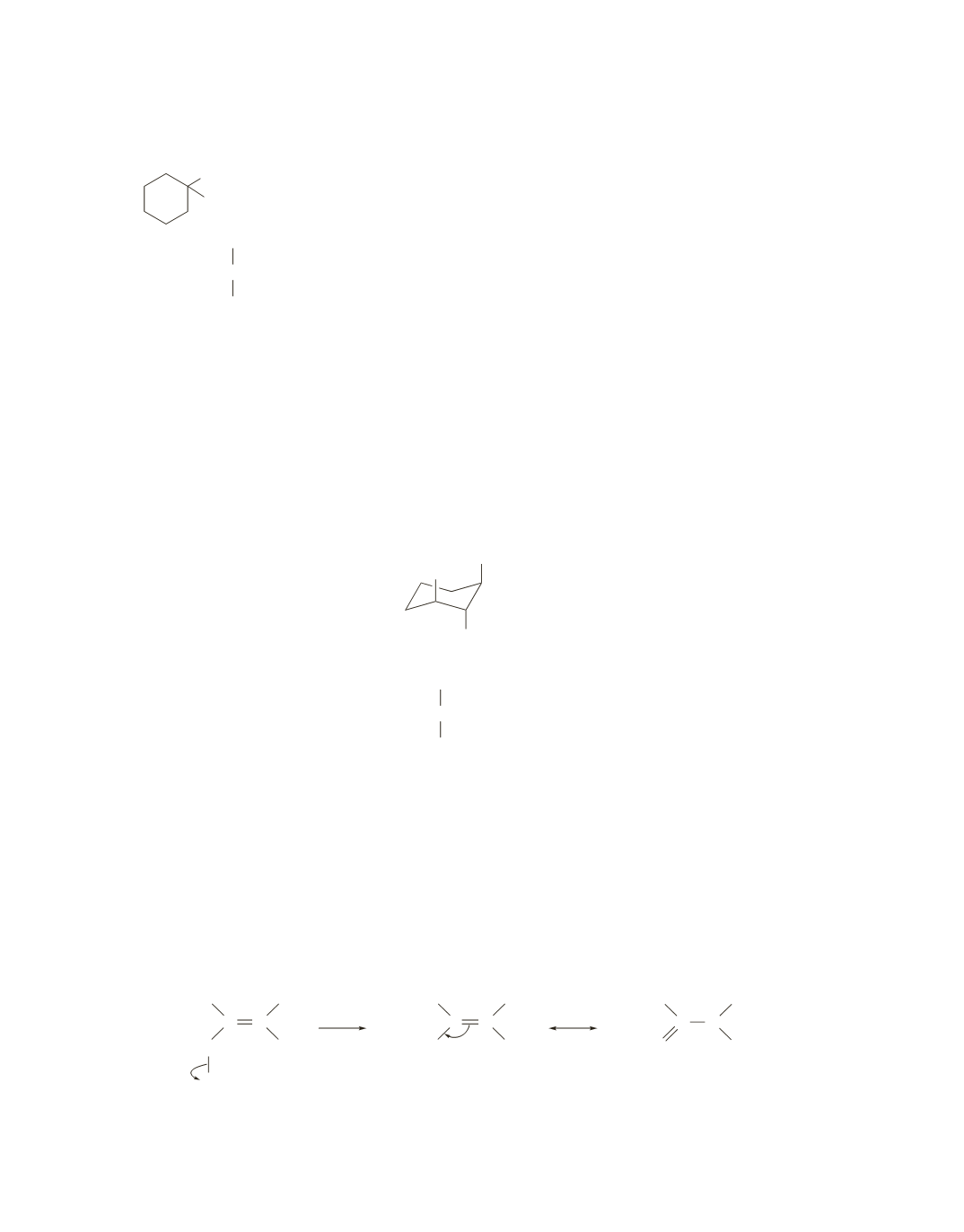
In order to undergo an E2 reaction, the substituents to be eliminated (H and Br) must both be in axial positions.
Drawing the compound in the chair conformation shows that when Br is in an axial position, neither of the
adjacent
b
-carbons has a hydrogen in an axial position, so an elimination reaction cannot take place.
CH
3
Br
CH
3
43.
CH
3
CCH
2
Br
CH
3
CH
3
1-bromo-2,2-dimethylpropane
a.
The bulky
tert-
butyl substituent blocks the back side of the carbon bonded to the bromine to
nucleophilic attack, making an
S
N
2
reaction difficult. An
S
N
1
reaction cannot occur because it requires
formation of an unstable primary carbocation.
b.
It cannot undergo an E2 reaction, because the
b
-carbon is not bonded to a hydrogen.
It cannot undergo an E1 reaction, because that requires the formation of a primary carbocation.
44.
a.
trans
-4-Bromo-2-hexene (the compound on the right) is more reactive, because the carbocation that is
formed is stabilized by electron delocalization. (It is a secondary allylic cation.) The other alkyl halide
is a secondary alkyl halide and does not undergo an
S
N
1
reaction.
CH
3
CH
2
CH H
CC
CH
3
H
Br
H
CC
CH
3
H
+
CH
3
CH
2
CH H
CC
CH
3
H
+
CH
3
CH
2
CH
















