
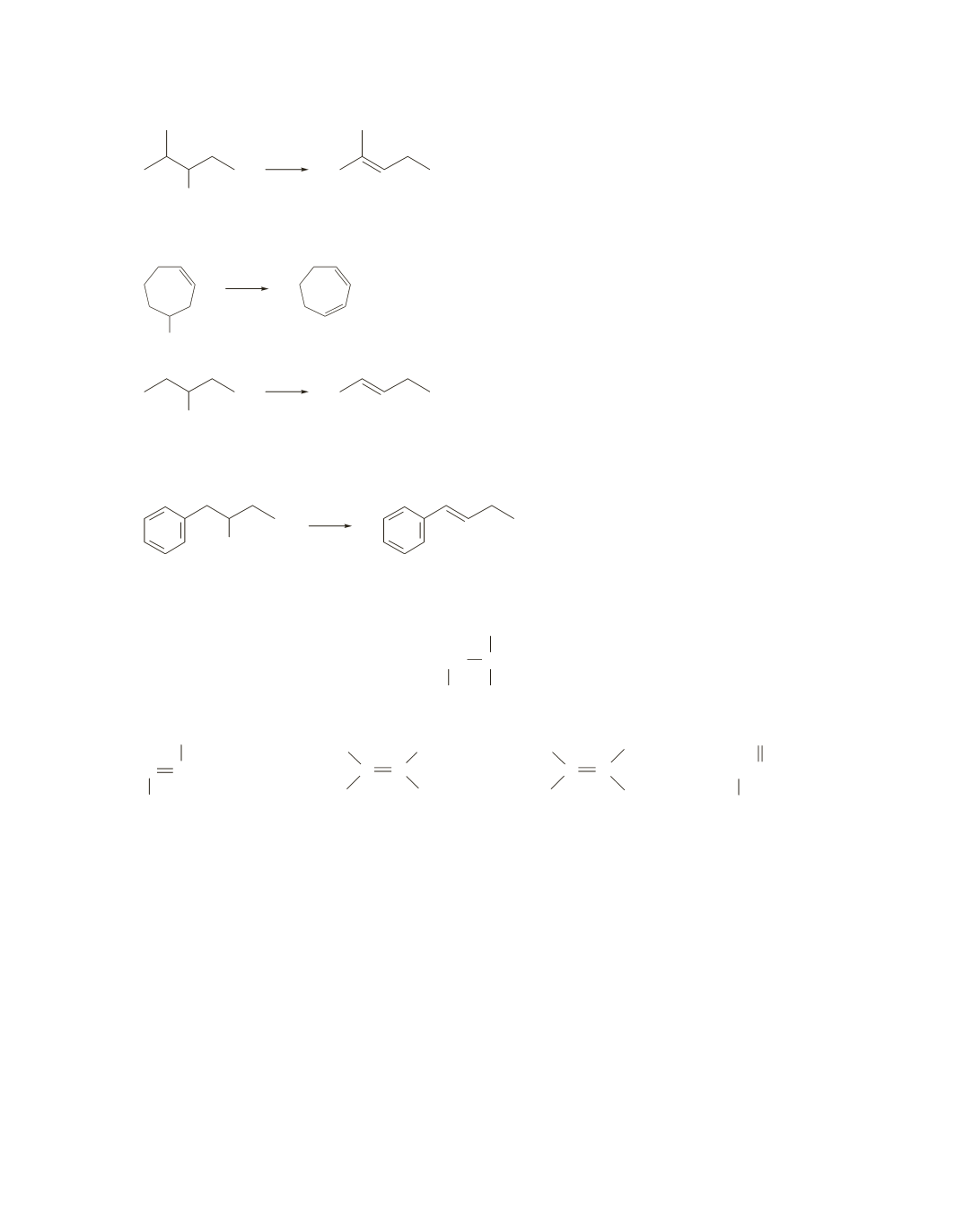
320 Chapter 9
Copyright © 2017 Pearson Education, Inc.
25.
a.
Br
It forms the more stable alkene (the alkene with the
most substituents bonded to the
sp
2
carbons), so it has
the more stable transition state.
b.
Br
It forms the more stable alkene (the double bonds are conjugated,
so it has the more stable transition state.
c.
Br
It has four hydrogens that can be removed to form
an alkene with two substituents on the
sp
2
carbons,
so it has a greater probability of having an effective
collision with the nucleophile than the other alkyl
halide that has only two such hydrogens.
d.
Br
It forms the more stable alkene (the new
double bond is conjugated with the phenyl
substituent), so it has the more stable
transition state.
26.
CH
3
C CCH
2
CH
3
CH
3
CH
3
>
CH
3
CCH
2
CH
3
Br
CH
3
CH
CH
3
3-bromo-2,3-dimethylpentane
>
CH
3
CHCCH
2
CH
3
CH
2
CH
3
(CH
3
)
2
CH
C C
H
3
C
CH
3
H
(CH
3
)
2
CH
C C
H
3
C
H
CH
3
>
Three alkyl substituents are
bonded to the
sp
2
carbons;
the largest groups are on
opposite sides of the
double bond.
Three alkyl substituents are
bonded to the
sp
2
carbons;
the largest groups are on the
same side of the double bond.
Two alkyl substituents
are bonded to the
carbons.
sp
2
Four alkyl substituents
are bonded to the
carbons.
sp
2
27.
The major product is the one predicted by Zaitsev’s rule, because the fluoride ion dissociates in the first
step, forming a carbocation. Loss of a proton from the carbocation follows Zaitsev’s rule, as it does in other
E1 reactions.
28.
a.
B because it forms the more stable carbocation.
b.
B because it forms the more stable alkene.
c.
B because it forms the more stable carbocation.
d.
A because it is less sterically hindered.
















