
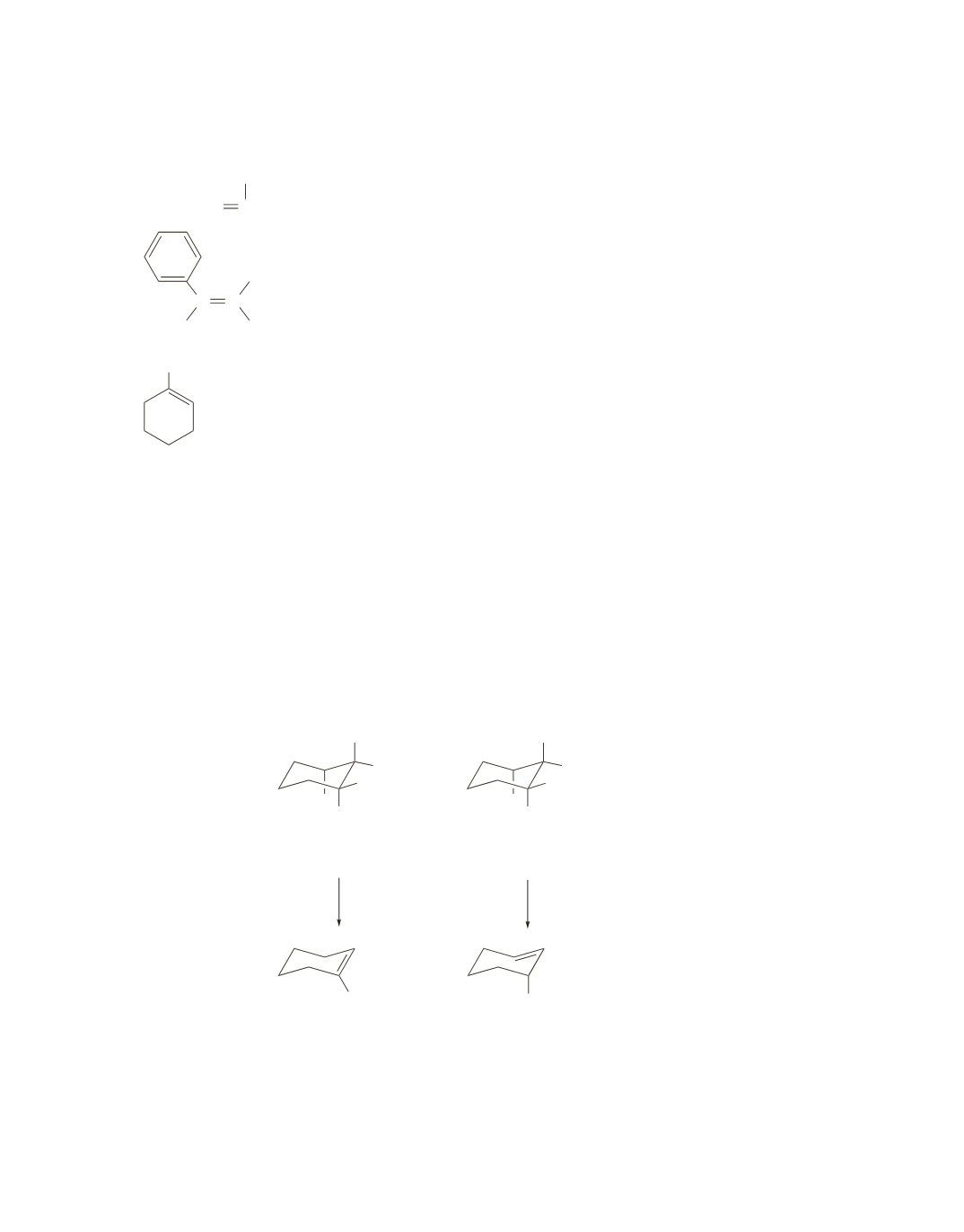
322 Chapter 9
Copyright © 2017 Pearson Education, Inc.
34.
Solved in the text.
35.
a.
CH
3
CH
2
CH CCH
3
CH
3
b.
C C
H
CH
3
CH
3
The larger substituent attached to one
sp
2
carbon and the larger
substituent attached to the other
sp
2
carbon are on opposite sides of the
double bond.
c.
CH
3
36.
E2 elimination reactions from six-membered rings occur only when the substituents to be eliminated are
both in axial positions.
In the cis isomer, when Br is in an axial position, there is an axial hydrogen on each of the adjacent
carbons. The one bonded to the same carbon as the ethyl group is more apt to be the one eliminated with
Br because the product formed is more stable and, therefore, more easily formed than the product formed
when the other H is eliminated with Br. (Recall that when there is a choice, a hydrogen is removed from
the
b
-carbon bonded to the fewest hydrogens.)
In the trans isomer, when Br is in an axial position, there is an axial hydrogen on only one adjacent carbon,
and it is not the carbon that is bonded to the ethyl group. Therefore, a different product is formed. (Notice
in this case there is no choice; there is only one hydrogen bonded by an axial bond to a
b
-carbon.)
H
Br
H
CH
2
CH
3
CH
2
CH
3
Br
H
H
CH
2
CH
3
CH
2
CH
3
1-ethylcyclohexene
3-ethylcyclohexene
H
H
cis-
1-bromo-2-ethylcyclohexane
trans
-1-bromo-2-ethylcyclohexane
















