
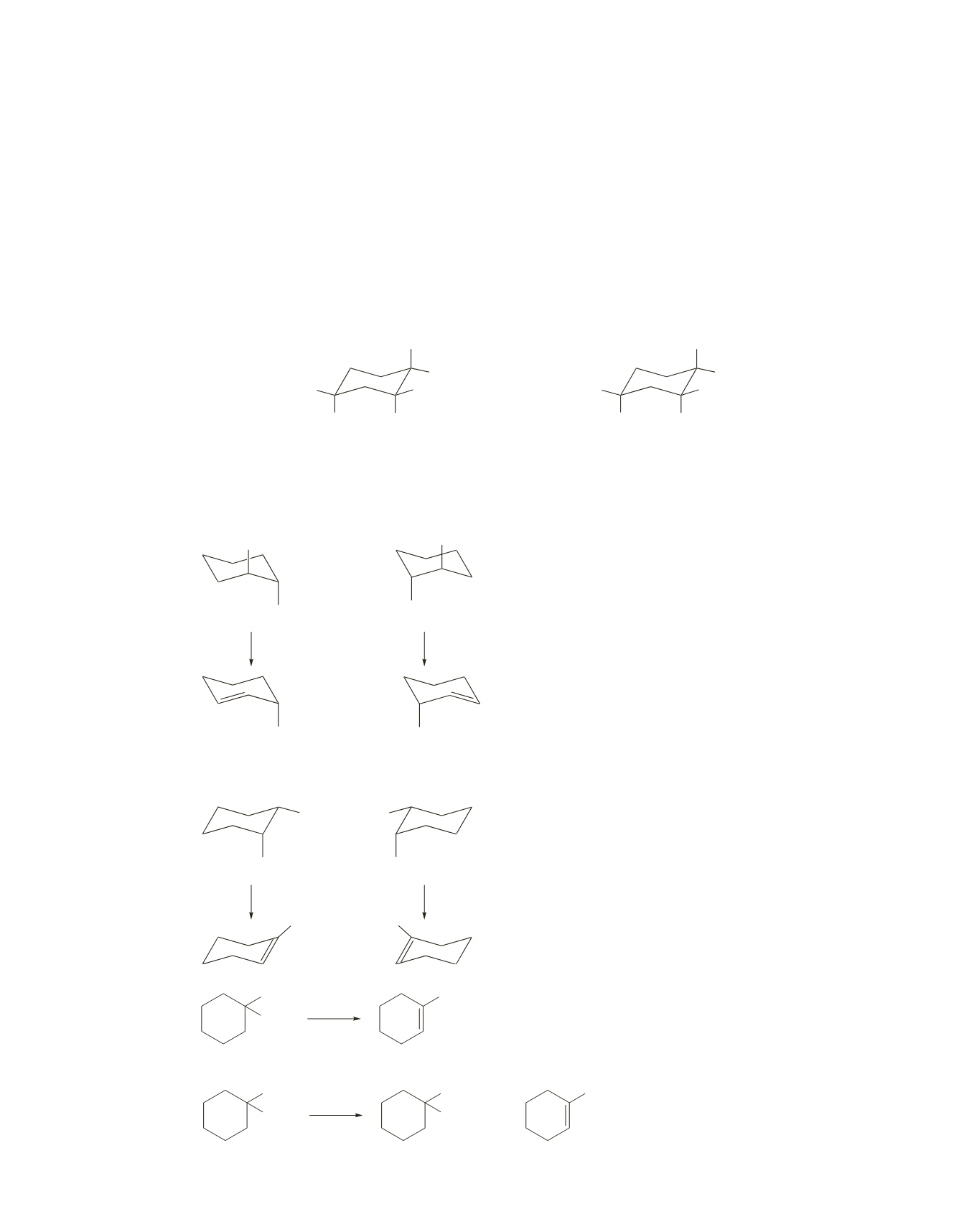
Chapter 9 323
Copyright © 2017 Pearson Education, Inc.
37.
In order for a six-membered ring to undergo an E2 reaction, the substituents that are to be eliminated must
both be in axial positions.
When bromine and an adjacent hydrogen are both in axial positions, the large
tert
-butyl substituent is in an
equatorial position in the cis isomer and in an axial position in the trans isomer. The rate constant for the
reaction is
k K
eq
.
Because a large substituent is more stable in an equatorial position than in an axial position, elimination
of the cis isomer occurs through its more stable chair conformer (
K
eq
is large; see page 427 of the text),
whereas elimination of the trans isomer has to occur through its less stable chair conformer (
K
eq
is small).
The cis isomer, therefore, reacts more rapidly in an E2 reaction.
H
Br
H
H
H
(CH
3
)
3
C
H
Br
H
H
(CH
3
)
3
C
H
cis
-1-bromo-4-
tert
-butylcyclohexane
trans
-1-bromo-4-
tert
-butylcyclohexane
38.
a.
trans-
1-Chloro-2-methylcyclohexane has two stereoisomers, and each forms an elimination product.
Notice that the elimination products are enantiomers.
CH
3
Cl
CH
3
CH
3
CH
3
Cl
Elimination occurs through
the less stable conformer.
CH
3
O
–
CH
3
O
–
b.
cis
-l-Chloro-2-methylcyclohexane has two stereoisomers, and each forms an elimination product.
Notice that the elimination products are enantiomers.
CH
3
Cl
CH
3
Cl
Elimination occurs through
the more stable conformer.
CH
3
O
–
CH
3
CH
3
O
–
CH
3
c.
CH
3
only product
Cl
CH
3
O
−
CH
3
d.
OCH
3
CH
3
CH
3
OH
CH
3
CH
3
Cl
+
















