
348 Chapter 9
Copyright © 2017 Pearson Education, Inc.
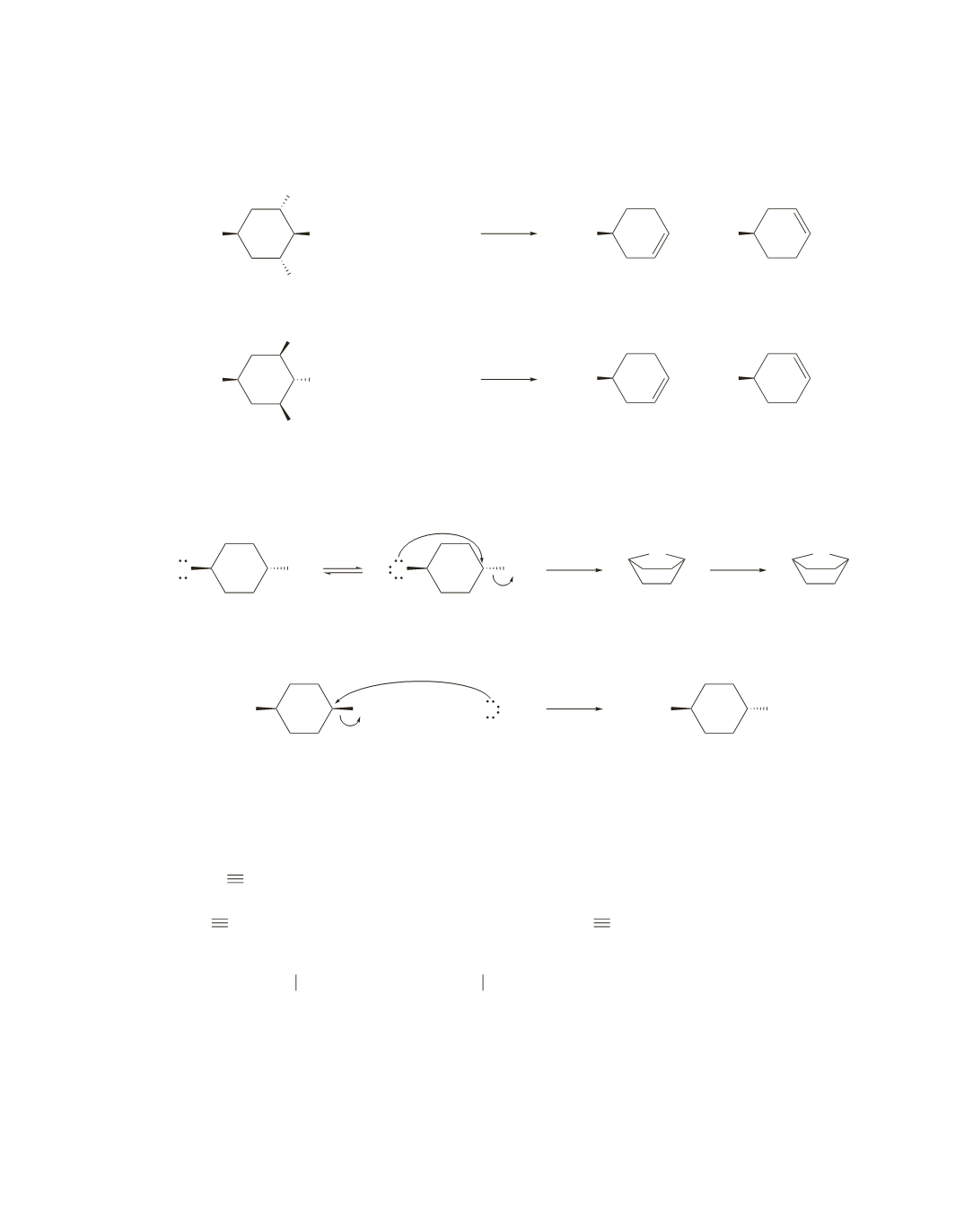
119.
a.
Both compounds form the same elimination products because they both have hydrogens bonded to the
same
b
-carbons that are anti to the bromine.
H
Br
HO
HO
−
+
+
HO
HO
+
HO
HO
cis
-4-bromocyclohexanol
H
Br
HO
HO
−
+
trans
-4-bromocyclohexanol
H
H
b.
Only the trans isomer can undergo an intramolecular substitution reaction because the
S
N
2
reaction
requires back-side attack.
HO
cis
-4-bromocyclohexanol
HO
+
HO
trans
-4-bromocyclohexanol
OH
Br
O
Br
H
O
HO
Br
HO
−
O
+
The cis isomer can undergo only an intermolecular substitution reaction.
−
HO
−
−
c.
The elimination reaction forms a pair of enantiomers because the reaction creates an asymmetric cen-
ter in the product. Both substitution reactions form a single stereoisomer, because the reaction does not
create an asymmetric center in the product.
120.
a.
1.
CH
3
CH
2
C CCH
2
CH
3
2.
CH
3
CH
2
CH
2
OCH
3
b.
CH
3
CH
2
C C
−
CH
3
CH
2
CH
2
CH
2
C
CH
3
CH
2
Br
C
−
CH
3
CH
2
CH
2
CH
2
Br
or
+
+
c.
CH
3
CH
2
O
−
CH
3
CHCH
2
CH
2
O
−
CH
3
CH
2
Br
CH
3
CHCH
2
CH
2
Br
CH
3
CH
3
or
+
+
















