
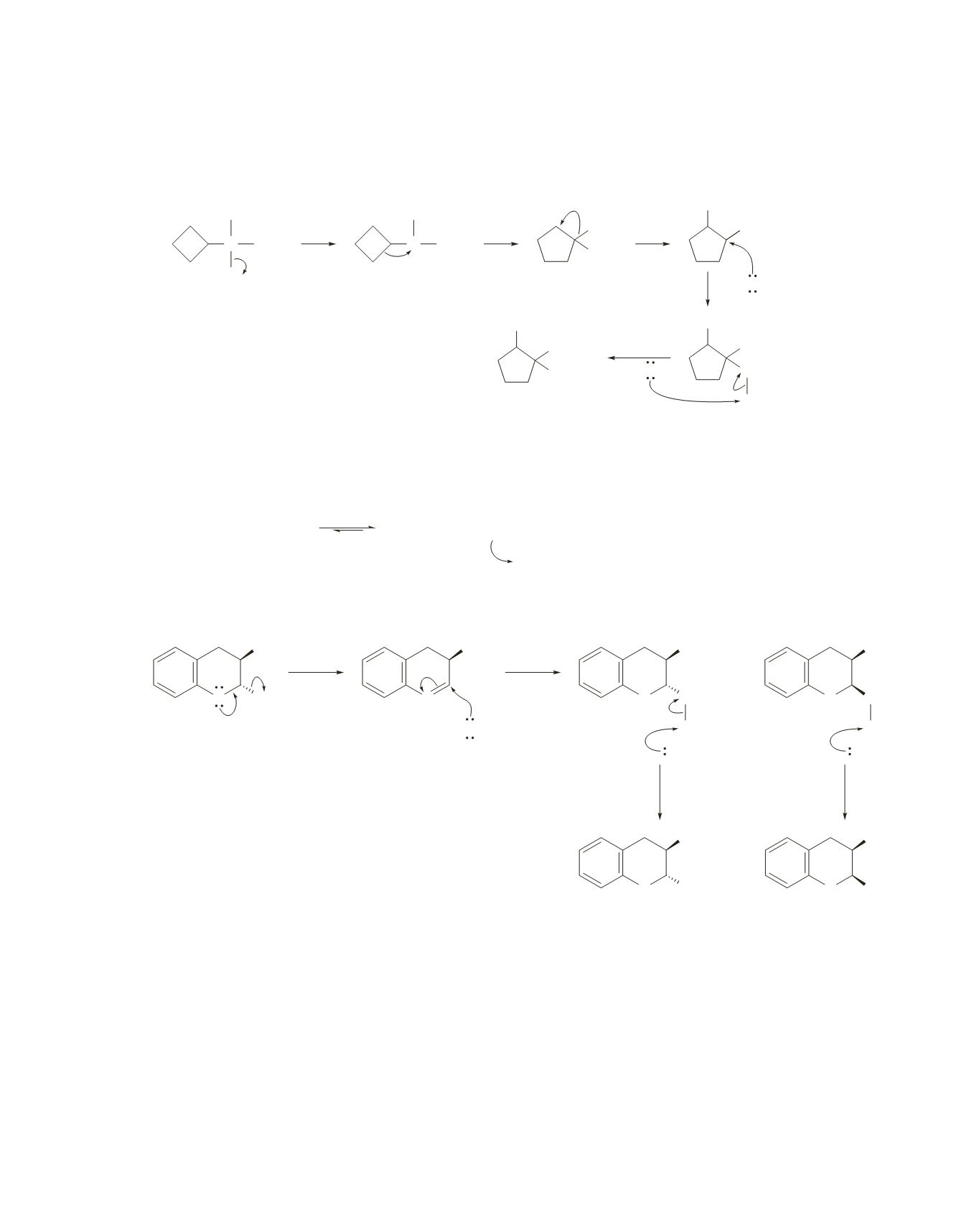
Chapter 9 349
Copyright © 2017 Pearson Education, Inc.
121.
A tertiary carbocation with a strained four-membered ring is less stable than a secondary carbocation with
an unstrained five-membered ring, so a carbocation rerrangement occurs. A second carbocation rearrange-
ment forms a tertiary carbocation.
C CH
3
CH
3
Cl
C CH
3
CH
3
CH
3
2 1
3
3
2
1
CH
3
Cl
−
CH
3
OCH
3
CH
3
CH
3
OH
CH
3
CH
3
CH
3
CH
3
OCH
3
H
CH
3
OH
H
CH
3
OH
+
+
+
+
+
+
122.
Tetrahydrofuran can solvate a charge better than diethyl ether can, because the floppy ethyl substituents
of diethyl ether provide steric hindrance, making it difficult for the nonbonding electrons of the oxygen to
approach the positive charge that is to be solvated.
123.
CH
3
CH
2
Cl
K
+
I
−
acetone
CH
3
CH
2
I
K
+
Cl
−
K
+
Cl
−
precipitates out
in acetone, which drives
the reaction to the right.
+
+
124.
a.
Br
Br
O
CH
3
OH
Br
O
Br
O OCH
3
H
B
H
OCH
3
Br
O
+
+
+
+
B
Br
O OCH
3
+
HB
+
+
HB
+
OCH
3
Br
O
+
b.
Two products are obtained because methanol can add to the top or bottom of the planar double bond.
c.
One bromine is eliminated with the help of one of oxygen’s lone pairs, forming a species in which
the positive charge is shared by a carbon and an oxygen. The oxygen cannot help eliminate the other
bromine.
















