
Chapter 9 351
Copyright © 2017 Pearson Education, Inc.
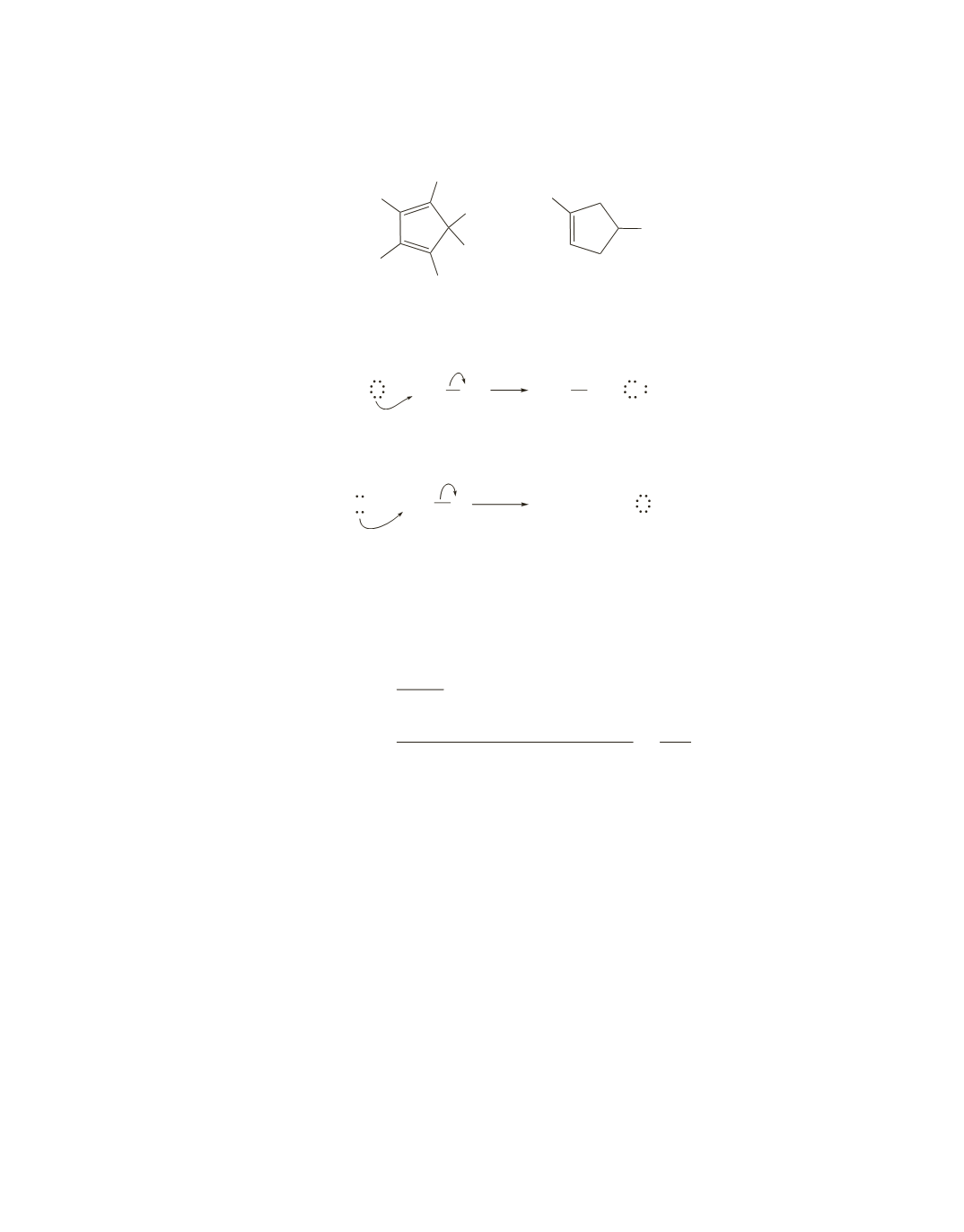
128.
A Diels–Alder reaction between hexachlorocyclopentadiene and 1,4-dichlorocyclopentene forms
chlordane.
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Cl
+
129.
I
-
is a good nucleophile in a polar solvent such as methanol, so it reacts rapidly with methyl bromide, causing
the concentration of
I
-
to decrease rapidly.
I
−
+
CH
3
Br
fast
CH
3
I
+
Br
−
I
-
is a good leaving group, so methyl iodide undergoes an
S
N
2
reaction with methanol.
Methanol is a poor nucleophile, so the
S
N
2
reaction is slow. Therefore, iodide ion returns slowly to its
original concentration.
CH
3
OH
+
CH
3
I
slow
CH
3
OCH
3
+
I
−
H
+
+
130.
It does not undergo an
S
N
2
reaction, because of steric hindrance to back-side attack.
It does not undergo an
S
N
1
reaction, because the carbocation that would be formed is unstable; the ring
structure prevents it from achieving the
120
°
bond angles required for an
sp
2
carbon.
131.
The equation needed to calculate
K
eq
from the change in free energy is given in Section 5.6.
ln
K
eq
=
-
∆
G
°
RT
ln
K
eq
=
-
1
-
21.7 kcal mol
-
1
2
0.001986 kcal mol
-
1
K
-
1
*
303 K
=
21.7
0.60
ln
K
eq
=
36.1
K
eq
=
4.8
*
10
15
As expected, this highly exergonic reaction has a very large equilibrium constant.
















