
346 Chapter 9
Copyright © 2017 Pearson Education, Inc.
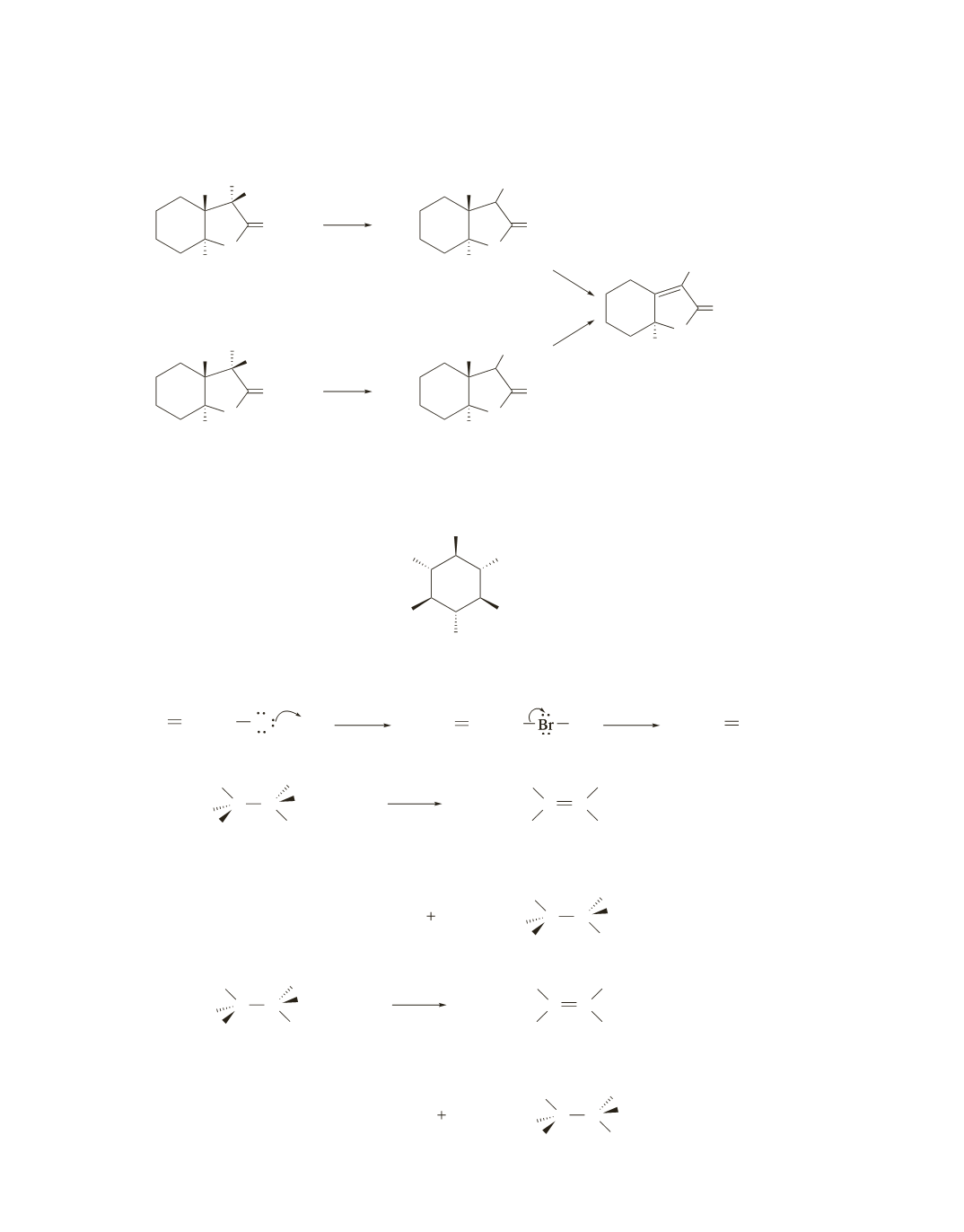
The two tertiary alkyl halides form the same product when they react with a weak base (an E1 reaction)
because both form the same carbocation.
O
H
H
Br
CH
3
O
O
H
H
O
CH
3
+
O
H
O
CH
3
a.
O
H
H
CH
3
Br
O
O
H
H
O
CH
3
+
b.
113.
In order to undergo an E2 reaction, a chlorine and a hydrogen bonded to an adjacent carbon must be trans to each
other so they can both be in the required axial positions. Every Cl in the following compound has a Cl trans to it, so
no Cl has a hydrogen trans to it. Therefore, it is the least reactive of the isomers; it cannot undergo an E2 reaction.
Cl
Cl
Cl
Cl
Cl
Cl
114.
The silver ion increases the ease of departure of the halogen atom by sharing one of bromine’s lone pairs,
which weakens the carbon–bromine bond.
CH
3
CH CHCH
2
Br
+
Ag
+
CH
3
CH CHCH
2
+
Ag
CH
3
CH CHCH
2
+
+
AgBr
115.
For a description of how to do this problem, see the Problem-Solving Strategy on page 425.
a.
H
CH
3
CH
2
CH
3
CH
2
CH
3
(
E
)-3-methyl-2-pentene
(3
S
,4
S
)-3-bromo-4-methylhexane
C C
H
CH
3
CH
2
CH
3
CH
2
CH
3
C C
Br
H
CH
3
O
–
CH
3
CH
CH
3
O
2
CH
3
CH
2 3
CH
C C
H
H
b.
CH
3
O
CH
3
CH
2
CH
3
CH
2 3
CH
C C
H
H
CH
3
CH
2
CH
3
CH
3
CH
2
CH
3
H
CH
3
CH
2
(
E
)-3-methyl-2-pentene
(3
R
,4
R
)-3-bromo-4-methylhexane
C C
H
CH
3
CH
2
C C
Br
H
CH
3
O
–
















