
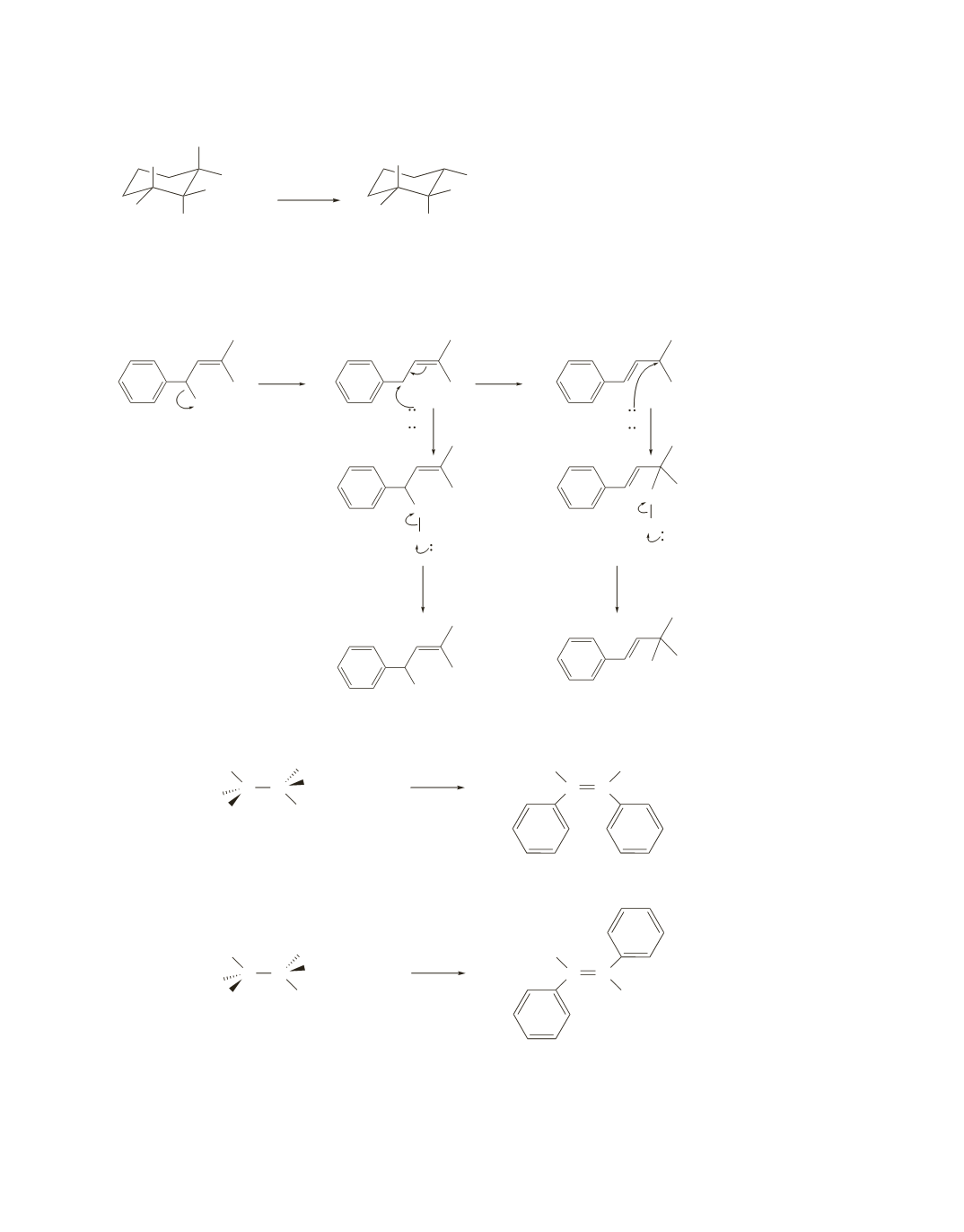
350 Chapter 9
Copyright © 2017 Pearson Education, Inc.
125.
CH
3
CH
3
H
CH
3
Cl
H
CH
3
H
OCH
3
S
N
2/E2
CH
3
O
−
H
H
There is only a substitution product.
The reactant does not undergo elimination because when Cl is in an
axial position, neither of the
b
-carbons is bonded to an axial hydrogen.
126.
127.
a.
H
CH
3
(
Z
)-3-methyl-2-pentene
(1
S
,2
S
)-1-bromo-1,2-diphenylpropane
C C
H
5
C
6
H
5
C
6
H
CH
3
C C
Br
H
b.
H
CH
3
(
E
)-3-methyl-2-pentene
(1
S
,2
R
)-1-bromo-1,2-diphenylpropane
C C
H
5
C
6
H
5
C
6
H
CH
3
C C
Br
H
Cl
OCH
H
B
OCH
HB
OCH
H
B
OCH
HB
CH OH
+
CH OH
3
3
3
3
3
3
+
+
+
+
+
















