
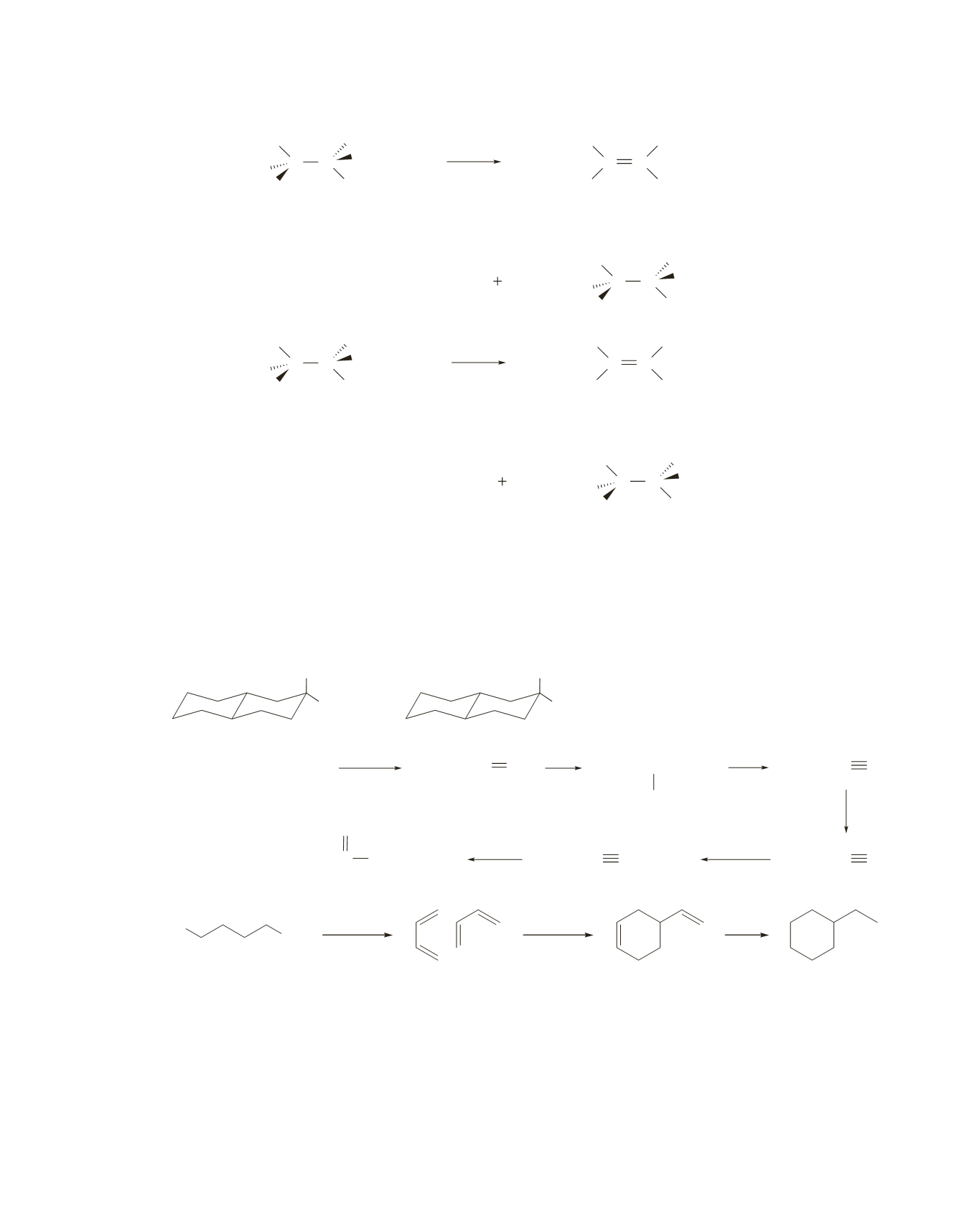
Chapter 9 347
Copyright © 2017 Pearson Education, Inc.
c.
CH
3
CH
2
CH
3
CH
3
CH
2
CH
3
H
CH
3
CH
2
(
Z
)-3-methyl-2-pentene
(3
S
,4
R
)-3-bromo-4-methylhexane
C C
H
CH
3
CH
2
C C
Br
H
CH
3
O
CH
3
CH
2
CH
3
CH
2 3
CH
C C
H
H
CH
3
O
–
d.
CH
3
CH
2
CH
3
CH
3
CH
2
CH
3
H
CH
3
CH
2
CH
3
CH
2
(
Z
)-3-methyl-2-pentene
(3
R
,4
S
)-3-bromo-4-methylhexane
C C
H
CH
3
CH
2
C C
Br
H
CH
3
O
CH
3
CH
2 3
CH
C C
H
H
CH
3
O
–
116.
a.
3 2
2
3 2
2
CH CH CD CH and
CH CH CH CH
b.
The deuterium-containing compound results from elimination of HBr, whereas the non-deuterium-
containing compound results from elimination of DBr. The deuterium-containing compound is
obtained in greater yield, because a C—H bond is easier to break than a C—D bond.
117.
CH
3
OCH
3
OCH
3
CH
3
+
118.
a.
Br
2
2
−
NH
2
tert
-BuO
−
CH
3
CH
2
CH
2
CH
2
Br
CH
3
CH
2
CH CH
2
CH
3
CH
2
CHCH
2
Br
Br
CH
3
CH
2
C CH
−
NH
2
CH
3
CH
2
C C
−
CH
3
CH
2
Br
CH
3
CH
2
C CCH
2
CH
3
H
2
O
CH
3
CH
2
C CH
2
CH
2
CH
3
H
2
SO
4
O
b.
Diels-Alder
reaction
One equivalent reacts
in the Diels–Alder
reaction as a diene and
the other as a dienophile.
H
2
Pd/C
tert
-BuO
−
excess
Br
Br
















