
450 Chapter 13
Copyright © 2017 Pearson Education, Inc.
b.
The absorption band at 1685 cm
-
1
indicates a carbon–oxygen double bond. The absence of a
strong and broad absorption band at 3000 cm
-
1
rules out the carboxylic acid, and the absence of an
absorption band at 2700 cm
-
1
rules out the aldehyde. Therefore, it must be one of the ketones. The
ketone with the conjugated carbonyl group would be expected to show a C
“
O stretch at 1685 cm
-
1
,
whereas the ketone with the isolated carbonyl group would show a C
“
O stretch at 1720 cm
-
1
.
Thus, the compound is the ketone with the conjugated carbonyl group.
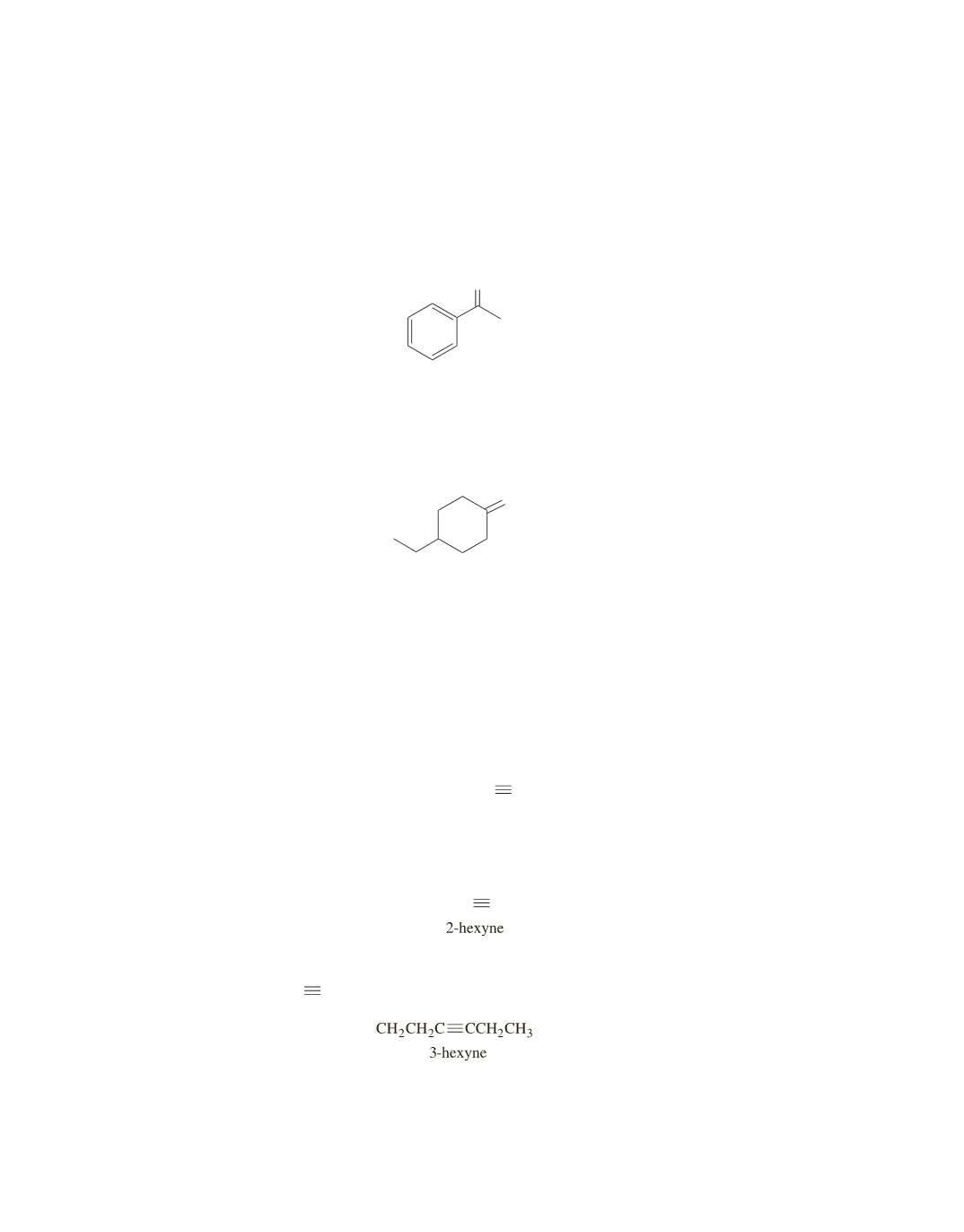
O
c
. The absorption band at 1700 cm
-
1
indicates a carbon–oxygen double bond. The absence of an
absorption band at 1600 cm
-
1
rules out the ketones with the benzene or cyclohexene rings. The
absence of absorption bands at 2100 cm
-
1
and 3300 cm
-
1
rules out the ketone with the carbon–
carbon triple bond. Therefore, it must be 4-ethylcyclohexanone.
O
63.
The absorption bands at 2700 cm
-
1
for the aldehyde hydrogen and at 1380 cm
-
1
for the methyl group
will distinguish the compounds.
A
will have the band at 2700 cm
-
1
but not the one at 1380 cm
-
1
.
B
will have the band at 1380 cm
-
1
but not the one at 2700 cm
-
1
.
C
will have the band at both 2700 cm
-
1
and 1380 cm
-
1
.
64.
1-Hexyne will show absorption bands at 3300 cm
-
1
for a hydrogen bonded to an
sp
carbon and at
2100 cm
-
1
for the triple bond.
CH
3
CH
2
CH
2
CH
2
C CH
1-hexyne
2-Hexyne will show the absorption band at 2100 cm
-
1
but not the one at 3300 cm
-
1
.
CH
3
CH
2
CH
2
C CCH
3
3-Hexyne will not show the absorption band at either 3300 cm
-
1
or 2100 cm
-
1
(there is no change in
dipole moment when the
C C
stretches).
















