
446 Chapter 13
Copyright © 2017 Pearson Education, Inc.
48.
Enovid would have its carbonyl stretch at a higher frequency. The carbonyl group in Norlutin has some
single-bond character because of electron delocalization as a result of having conjugated double bonds.
The single-bond character causes the carbon–oxygen bond to be easier to stretch than the carbon–oxygen
bond in Enovid, which has isolated double bonds and, therefore, no electron delocalization.
O
Norlutin
−
O
+
49.
OH 3300–3000
NH 3600–3200
sp
CH 3300
sp
2
CH 3050
sp
3
CH 2950
C C
C N
O
RCH 2700
2100
2250
C O
C C
1700
1600
1500
C O
C N
1250–1050
1230–1030
3600 cm
−
1
3000
1800
1400
1000
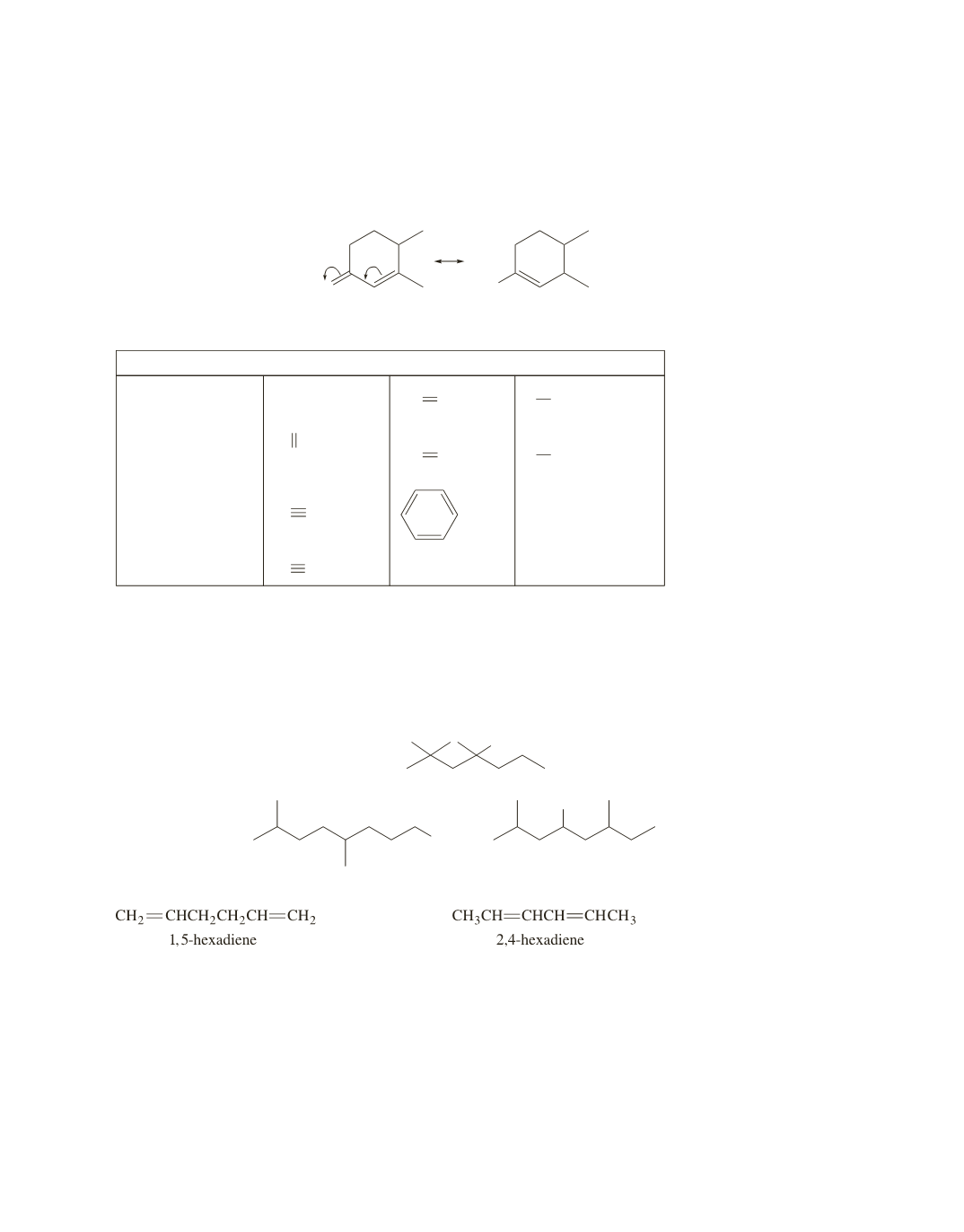
50.
The molecular weight of each of the alcohols is 158. The peak at
m
>
z
=
140 is due to loss of water
(158
-
18); each of the alcohols will show such a peak. The peaks at
m
>
z
=
87, 115, and 143 are due to
loss of a group with five carbons (C
5
H
11
), a group with three carbons (C
3
H
7
), and a methyl group, respec-
tively. Only 2,2,4-trimethyl-4-heptanol can lose all three groups via
a
-cleavage.
OH
OH
OH
51.
One way to distinguish the two compounds is by the presence or absence of an absorption band at
1370 cm
-
1
due to the methyl group that 2,4-hexadiene has but that 1,5-hexadiene does not have. In
addition, 2,4-hexadiene has conjugated double bonds; therefore, its double bonds have some single-bond
character due to electron delocalization. Consequently, they are easier to stretch than the isolated double
bonds of 1,5-hexadiene. Therefore, the carbon–carbon double-bond stretch of 2,4-hexadiene will be at a
smaller wavenumber than the carbon–carbon double-bond stretch of 1,5-hexadiene.
















