
Chapter 13 447
Copyright © 2017 Pearson Education, Inc.
52.
The fact that the abundance of the M
+
2 peak is 30% of the abundance of the M peak indicates that the com-
pound has one chlorine atom. The peak at
m
>
z
=
77 is due to loss of the chlorine atom (112
-
35
=
77).
The fact that the peak at
m
>
z
=
77 does not fragment indicates that it is a phenyl cation. Therefore, the
compound is chlorobenzene.
Cl
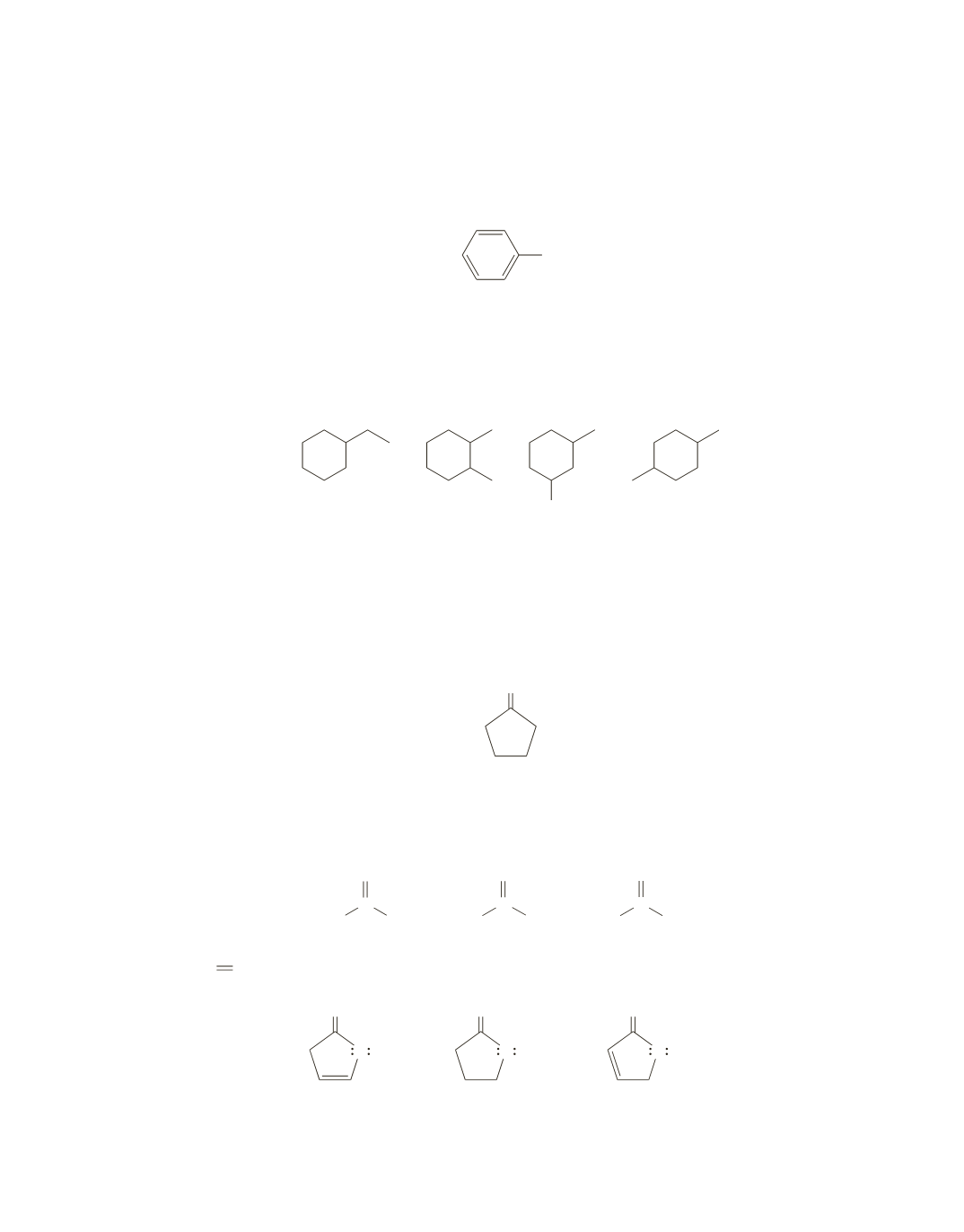
53.
Dividing 112 by 13 gives 8 with 8 left over, giving a base value of C
8
H
16
, and because the compound is a
hydrocarbon, this is also its molecular formula. The molecular formula indicates that it has one degree of
unsaturation, which is accounted for by the fact that we know it has a six-membered ring. Possible struc-
tures are shown here. Possible stereoisomers are not shown: the second and third structures have three
stereoisomers, and the fourth structure has two stereoisomers.
54.
The absorption band at 1740 cm
-
1
indicates that the compound has a carbonyl group, and the absence
of an absorption band at 1380 cm
-
1
indicates that it has no methyl groups. The absence of an absorption
band at 1600 cm
-
1
indicates that the compound does not have a carbon–carbon double bond, and the
absence of an absorption band at 3050 cm
-
1
indicates that the compound does not have hydrogens
bonded to
sp
2
carbons.
From the molecular formula, you can deduce that the compound is
cyclopentanone
.
O
55.
Hydrogens are more electron-withdrawing than alkyl groups. Therefore, the carbonyl group bonded to
two relatively electron-withdrawing hydrogens has the largest wavenumber for its C
“
O absorption band,
whereas the carbonyl group bonded to two alkyl groups has the lowest wavenumber.
O
C
H H
>
O
C
H
3
C
H
>
O
C
H
3
C
CH
3
56.
The
C O
absorption band of the three compounds decreases in the following order.
O
O
O
O
O
O
>
>
















