
Chapter 13 453
Copyright © 2017 Pearson Education, Inc.
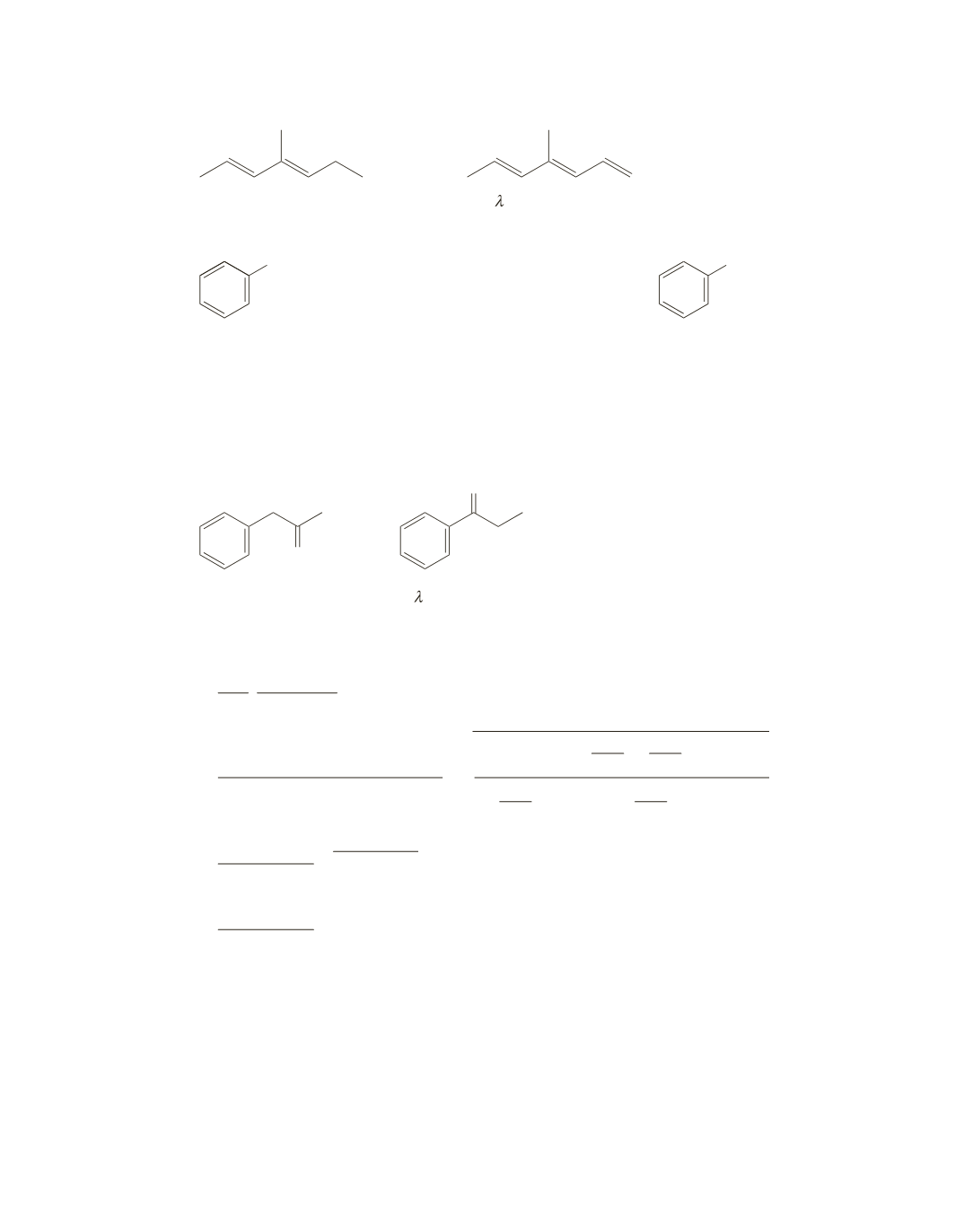
72.
a.
and
The
max
will be at a longer wavelength,
because there are three conjugated double bonds.
b.
OH
and
OCH
3
The
l
max
of the phenolate ion is at a lon-
ger wavelength than the
l
max
of phenol.
Because the p
K
a
of phenol is 10, the
l
max
will be at a longer wavelength at
pH
=
11 than at pH
=
7.
The
l
max
is pH-independent, so it will be the
same at pH 7 and 11.
c.
and
The
max
will be at a longer wavelength
because the carbonyl group is conjugated
with the benzene ring.
O
O
73.
v
∼
=
1
2
p
c
c
f
(
m
1
+
m
2
)
m
1
m
2
d
1
>
2
v
∼
=
1
2
*
3.1416
*
3
*
10
10
cm s
-
1
a
10
*
10
5
g s
-
2
a
12
6.02
+
12
6.02
b
*
10
-
23
g
12
6.02
*
10
-
23
g
*
12
6.02
*
10
-
23
g
v
∼
=
1
18.85
*
10
10
2
10.0
*
10
28
v
∼
=
1
18.85
*
10
10
*
3.16
*
10
14
v
∼
=
1676 cm
-
1
















