
454 Chapter 13
Copyright © 2017 Pearson Education, Inc.
74.
a.
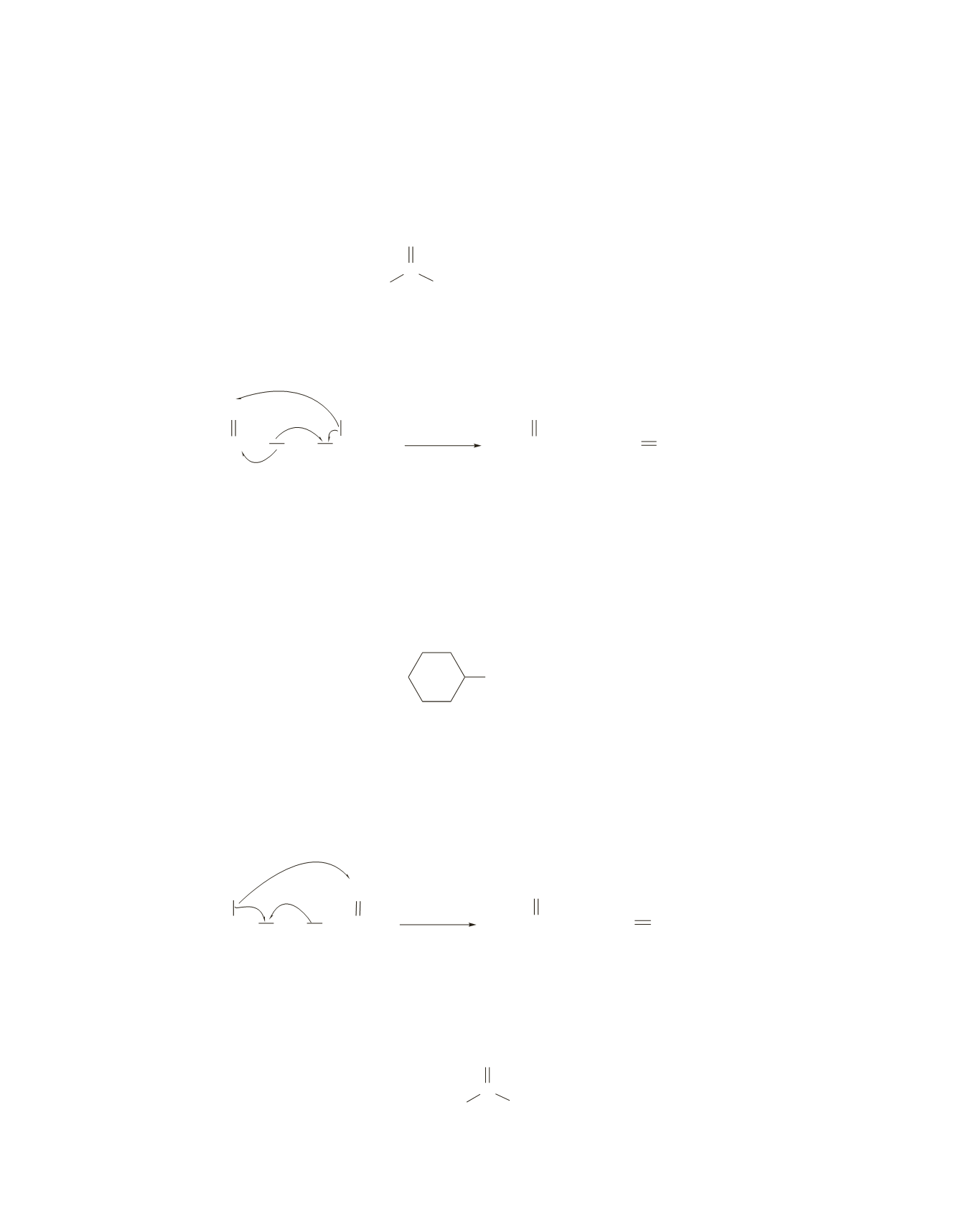
The IR spectrum indicates that the compound is an aliphatic ketone with at least one methyl group.
The M peak at
m
>
z
=
100 indicates that the ketone is a hexanone. The peak at 43 (100
-
57) for loss
of a butyl radical and the peak at 85 for loss of a methyl radical (100
-
15) suggest that the compound
is
2-hexanone
.
O
C
H
3
C
CH
2
CH
2
CH
2
CH
3
This is confirmed by the peak at 58 for loss of propene (100
-
42) as a result of a McLafferty
rearrangement.
CH
3
CCH
2
O
CH
2
CHCH
3
H
••
•
+
McLafferty
rearrangement
CH
3
CCH
2
OH
••
+
•
m
/
z
=
58
CH
2
CHCH
3
+
b.
The equal heights of the M and M
+
2 peaks at 162 and 164 indicate that the compound contains
bromine. The peak at
m
>
z
=
83 (162
-
79) is for the carbocation that is formed when the bromine
atom is eliminated. The IR spectrum does not indicate the presence of any functional groups, and it
shows that no methyl groups are present. The
m
>
z
peak
=
83 indicates a carbocation with a formula
of C
6
H
11
. The fact that the compound does not contain a methyl group indicates that the compound is
bromocyclohexane
.
Br
bromocyclohexane
c.
The absorption bands at 1700 cm
-
1
and 2700 cm
-
1
indicate that the compound is an aldehyde. The
molecular ion peak at
m
>
z
=
72 indicates that the aldehyde contains four carbons (C
4
H
8
O). The peak
at
m
>
z
=
44 for loss of a group with molecular weight 28 indicates that ethene has been lost as a result
of a McLafferty rearrangement.
CH
2
McLafferty
rearrangement
CH
2
CH
OH
••
+
•
m
/
z
=
44
CH
2
CH
2
CH
2
CH
2
CH
O
••
•
H
+
+
A McLafferty rearrangement can occur only if the aldehyde has a
g
-hydrogen. The only four-carbon
aldehyde that has a
g
-hydrogen is
butanal
.
O
C
CH
3
CH
2
CH
2
H
















