
456 Chapter 13
Copyright © 2017 Pearson Education, Inc.
5.
How can you distinguish between the IR spectra of the following compounds?
a.
OH
OH
and
b.
H
O
O
and
c.
OH
O
OCH
3
O
and
d.
O
OCH
3
and
e.
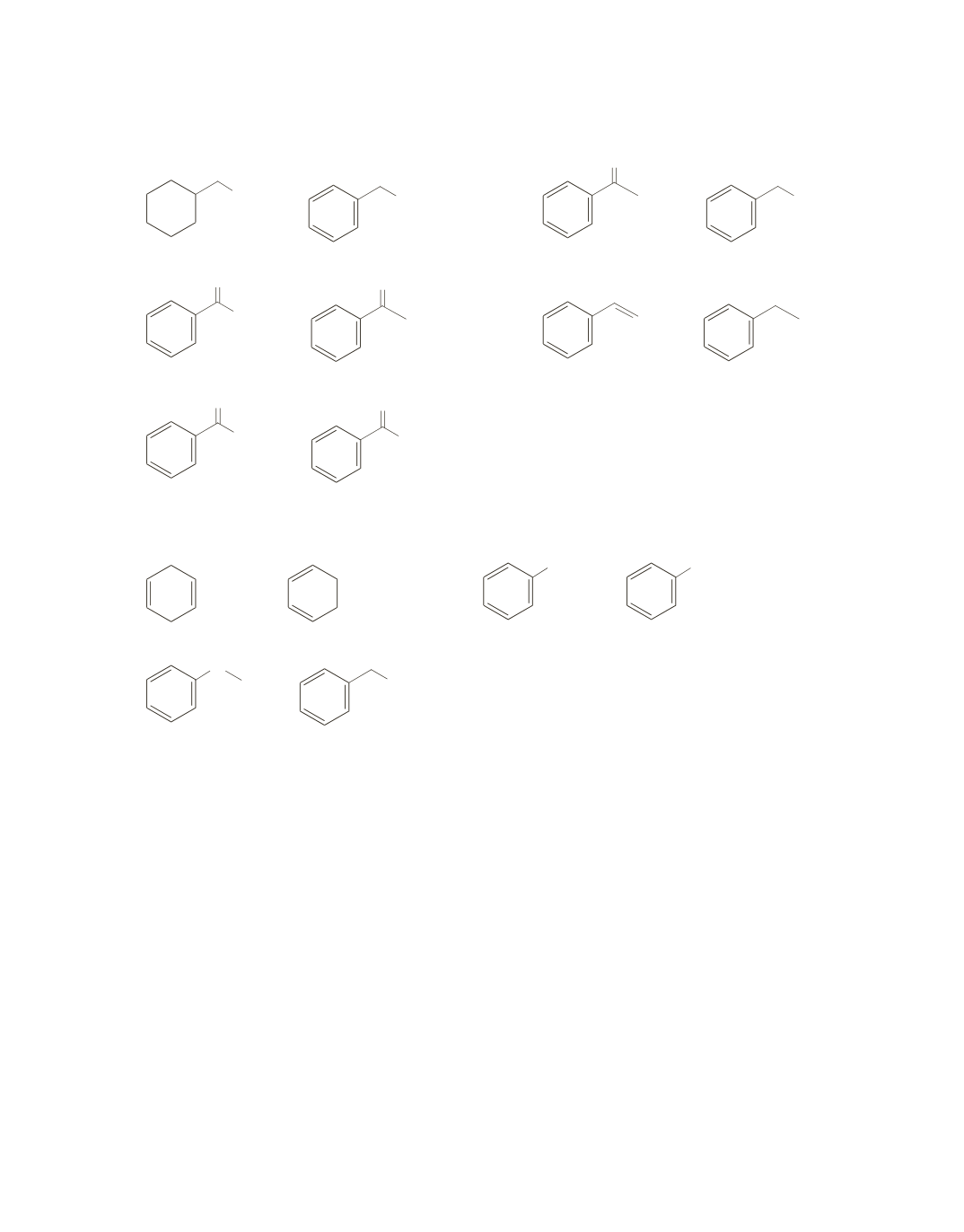
and
6.
Which compound has the greater
l
max
? (300 nm is a greater
l
max
than 250 nm.)
a.
and
b.
N
NH
2
and
H
c.
+
NH
2
NH
3
and
7.
A solution of a compound with a molar absorptivity of 1200 M
-
1
cm
-
1
at 297 nm gives an absorbance of
0.76 at that wavelength in a 1.0 cm quartz cell. What is the concentration of the solution?
8
Draw possible structures for an alcohol that has a molecular ion with an
m
>
z
value of 60.
9.
A bond between a carbon and an atom of similar electronegativity breaks ___________, whereas a bond
between a carbon and a more electronegative atom breaks _____________.
















