
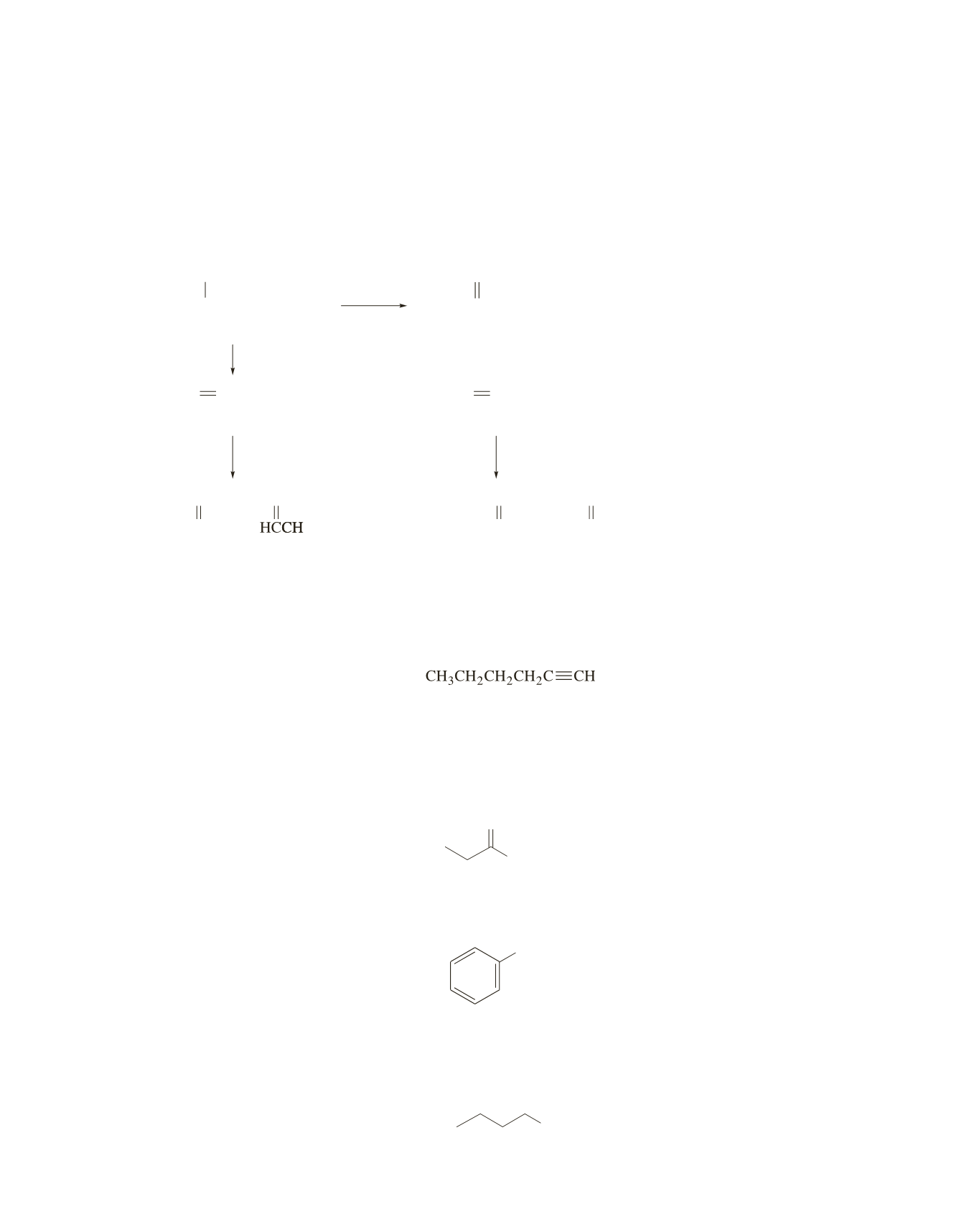
Chapter 13 449
Copyright © 2017 Pearson Education, Inc.
59.
The broad absorption band at 3300 cm
-
1
indicates that the compound has an OH group. The absence
of absorbance at 1700 cm
-
1
shows that the compound does not have a carbonyl group. The absence of
absorption at 2950 cm
-
1
indicates that the compound does not have any hydrogens bonded to
sp
3
carbons. Therefore, the compound is
phenol
.
60.
CH
3
CCH
2
CH
2 3
CH
H
2
SO
4
A
B
D
+
+
∇
oxidation
O
C
CH
2
CHCH
2
CH
2
CH
3
CH CH
3
CHCH
2
CH
3
CH
3
CHCH
2
CH
2
CH
3
OH
1.
,
_
78 ºC O
3
2. (CH ) S
3 2
1.
,
_
78 ºC O
3
2. (CH ) S
3 2
O
HCH
O
E
F
G
2
CH
2
CH
3
CH
3
CH
2
+
O
CH
O
CH
3
CH
61.
a.
The absorption band at 2100 cm
-
1
indicates a carbon–carbon triple bond, and the absorption band at
3300 cm
-
1
indicates a hydrogen bonded to an
sp
carbon.
b.
The absence of an absorption band at 2700 cm
-
1
indicates that the compound is not an aldehyde, and
the absence of a broad absorption band in the vicinity of 3000 cm
-
1
indicates that the compound is not
a carboxylic acid. The ester and the ketone can be distinguished by the absorption band at 1200 cm
-
1
that indicates the carbon–oxygen single bond of an ester.
O
OCH
2
CH
3
c.
The absorption band at 1360 cm
-
1
indicates the presence of a methyl group.
C(CH
3
)
3
62.
a.
The broad absorption band at 3300 cm
-
1
is characteristic of the oxygen–hydrogen stretch of an
alcohol; the absence of absorption bands at 1600 cm
-
1
and 3100 cm
-
1
indicates that it is not the
alcohol with a carbon–carbon double bond.
OH
















