
444 Chapter 13
Copyright © 2017 Pearson Education, Inc.
a.
3-Methylpentane is more apt to lose an ethyl radical (forming a secondary carbocation and a primary
radical) than a methyl radical (forming a secondary carbocation and a methyl radical). In addition,
3-methylpentane has two pathways to lose an ethyl radical. Therefore, the peak at
m
>
z
=
57 is more
intense than the peak at
m
>
z
=
71.
CH
3
CH
2
CHCH
2
CH
3
CH
3
3-methylpentane
b.
2-Methylpentane has two pathways to lose a methyl radical (forming a secondary carbocation and a
methyl radical in each pathway), and it cannot form a secondary carbocation by losing an ethyl radical.
(Loss of an ethyl radical would form a primary carbocation and a primary radical.) Therefore, it is
more apt to lose a methyl radical than an ethyl radical, so the peak at
m
>
z
=
71 is more intense than
the peak at
m
>
z
=
57.
CH
3
CHCH
2
CH
2
CH
3
2-methylpentane
CH
3
42.
1.
the change in the dipole moment when the bond stretches or bends
2.
the number of bonds that cause the absorption band
3.
the concentration of the sample
43.
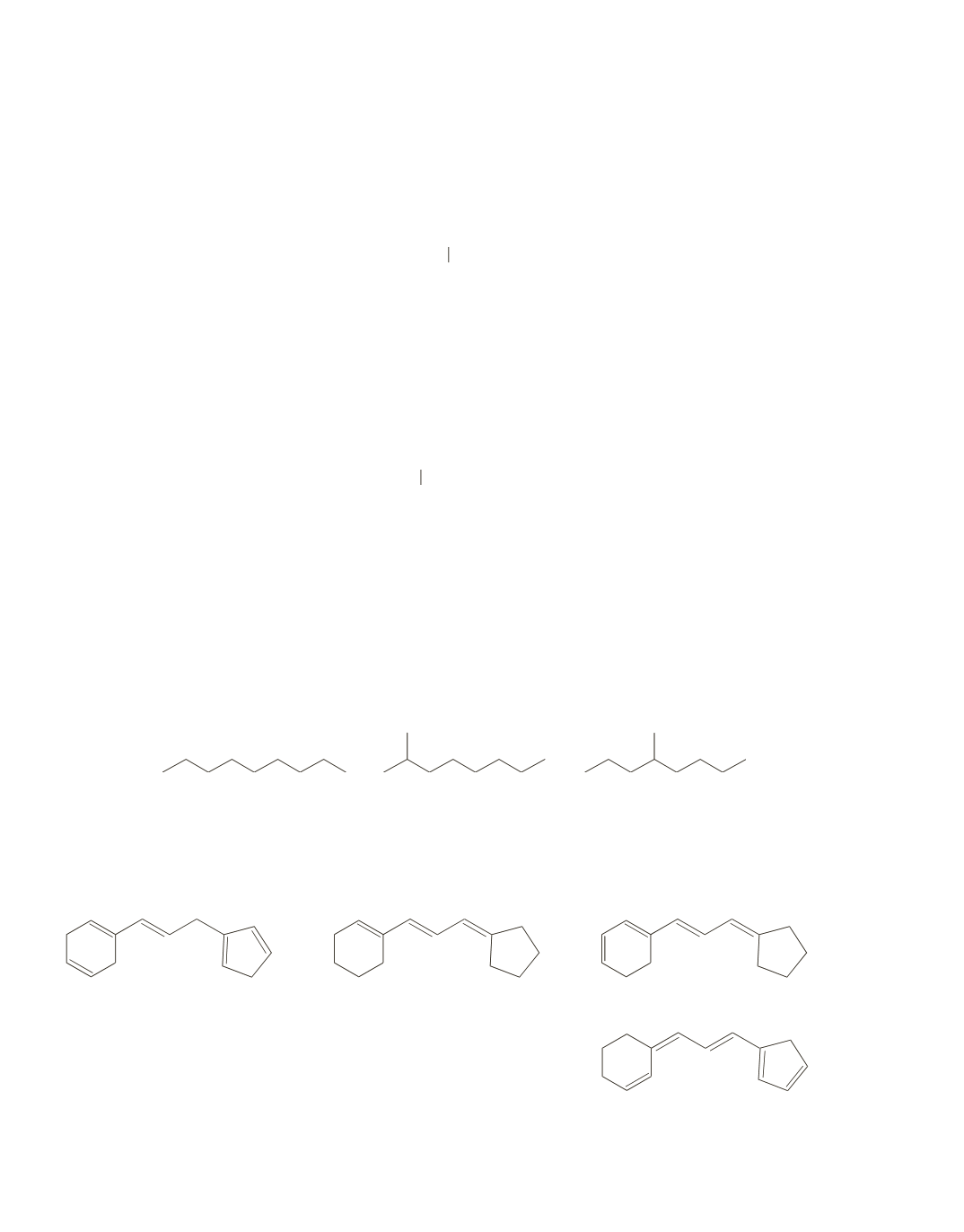
Dividing 128 by 13 gives 9 with 11 left over, giving a base value of C
9
H
20
. Because the compound is a
saturated hydrocarbon, we know that the base value is also the molecular formula of the compound. Some
possible structures are:
44.
The more conjugated double bonds in a compound, the greater its
l
max
.
C
, with 2 conjugated double bonds, has the smallest
l
max
.
D
, with 5 conjugated double bonds, has the greatest
l
max
.
C
<
D
B
<
A
<
















