
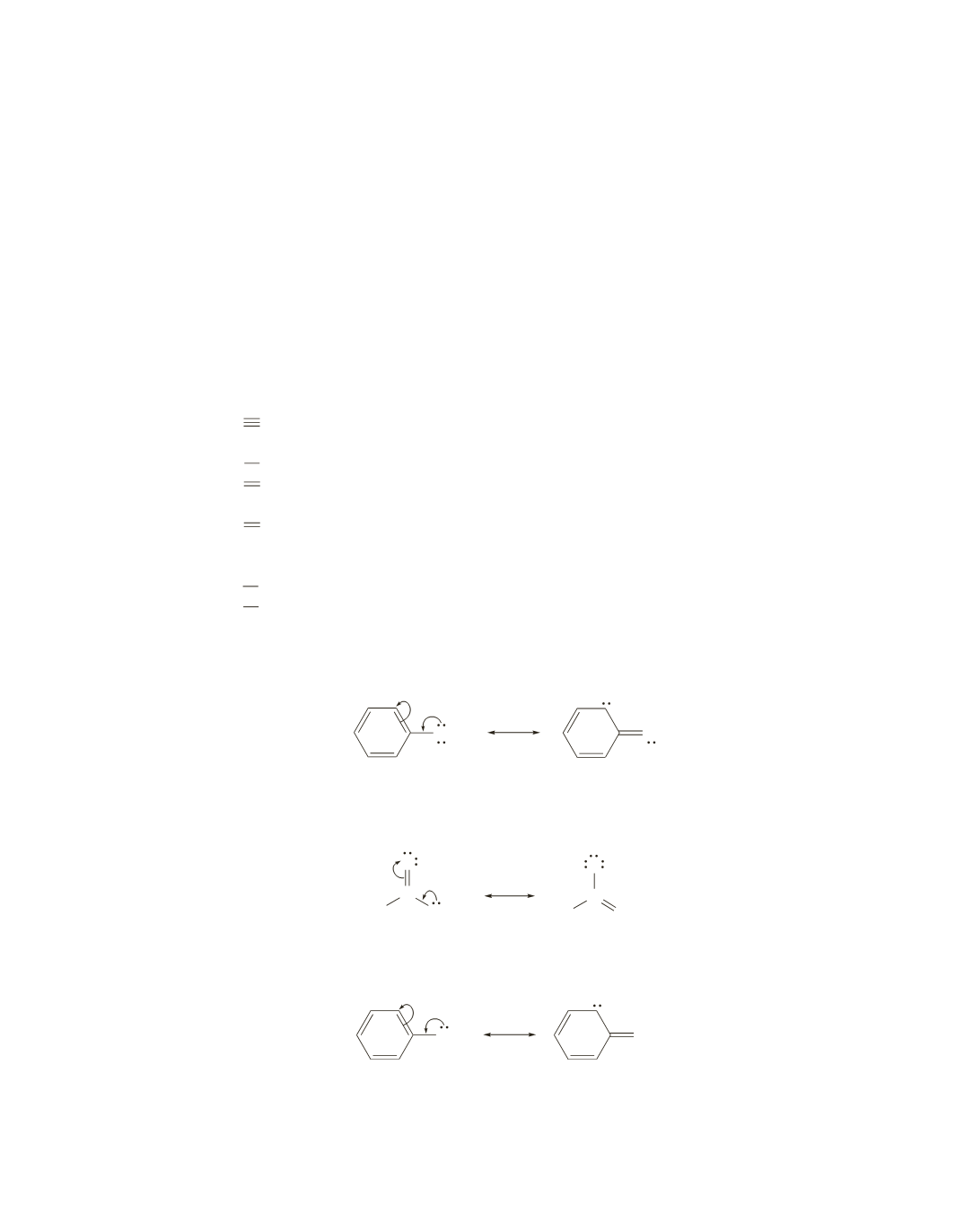
Chapter 13 439
Copyright © 2017 Pearson Education, Inc.
Spectrum
a.
shows a base peak at
m
>
z
=
59 due to loss of an ethyl radical (88
-
29), indicating that it is
the spectrum of 3-pentanol.
Spectrum
b.
shows a base peak at
m
>
z
=
45 due to loss of a propyl radical (88
-
43), indicating that it is
the spectrum of 2-pentanol.
19.
a.
2000 cm
-
1
(The larger the wavenumber, the higher the energy.)
b.
850 nm (The shorter the wavelength, the higher the energy.)
20.
The wavelength is the distance from the top of one wave to the top of the next wave. We see that
A
has a
longer wavelength than
B
.
Infrared radiation has longer wavelengths than visible light because infrared radiation is lower in energy.
Therefore,
A
depicts infrared radiation and
B
depicts visible light.
21.
a. 1.
C C
stretch A triple bond is stronger than a double bond, so it takes more energy to stretch
a triple bond.
2.
C H
stretch It requires more energy to stretch a given bond than to bend it.
3.
C N
stretch A double bond is stronger than a single bond, so it takes more energy to stretch
a double bond.
4.
C O
stretch A double bond is stronger than a single bond, so it takes more energy to stretch
a double bond.
b. 1.
C O
Vibrations of lighter atoms occur at larger wavenumbers.
2.
C C
Vibrations of lighter atoms occur at larger wavenumbers.
22.
a.
The carbon–oxygen stretch of phenol because it has partial double-bond character as a result of
electron delocalization.
OH
OH
+
−
b.
The carbon–oxygen double-bond stretch of a ketone because it has more double-bond character.
The double-bond character of the carbonyl group of an amide is reduced by electron delocalization.
R NH
2
C
O
R NH
2
C
O
+
−
c.
The C
¬
N stretch of aniline because it has partial double-bond character.
NH
2
NH
2
+
−
















