
434 Chapter 13
Copyright © 2017 Pearson Education, Inc.
9.
The ratio is 1:2:1.
To get the M peak, both Br atoms must be
79
Br. To get the M
+
4 peak, they both must be
81
Br. To get the
M
+
2 peak, the first Br atom can be
79
Br and the second
81
Br, or the first can be
81
Br and the second
81
Br.
So the relative intensity of the M
+
2 peak will be twice that of the others.
M
M
+
2
M
+
4
79
Br
79
Br
79
Br
81
Br
81
Br
81
Br
81
Br
79
Br
10.
The calculated exact masses show that only C
6
H
14
has an exact mass of 86.10955.
C
6
H
14
6(12.00000)
=
72.00000
C
4
H
6
O
2
4(12.00000)
=
48.00000
14(1.007825)
=
14.10955
6(1.007825)
=
6.04695
86.10955
2(15.9949)
=
31.9898
86.03675
C
4
H
10
N
2
4(12.00000)
=
48.00000
10(1.007825)
=
10.07825
2(14.0031)
=
28.0064
86.08465
11.
a.
A low-resolution spectrometer cannot distinguish between them because they both have the same
molecular mass (29).
b.
A high-resolution spectrometer can distinguish between these ions because they have different exact
molecular masses; one has an exact molecular mass of 29.039125, and the other an exact molecular
mass of 29.002725.
12.
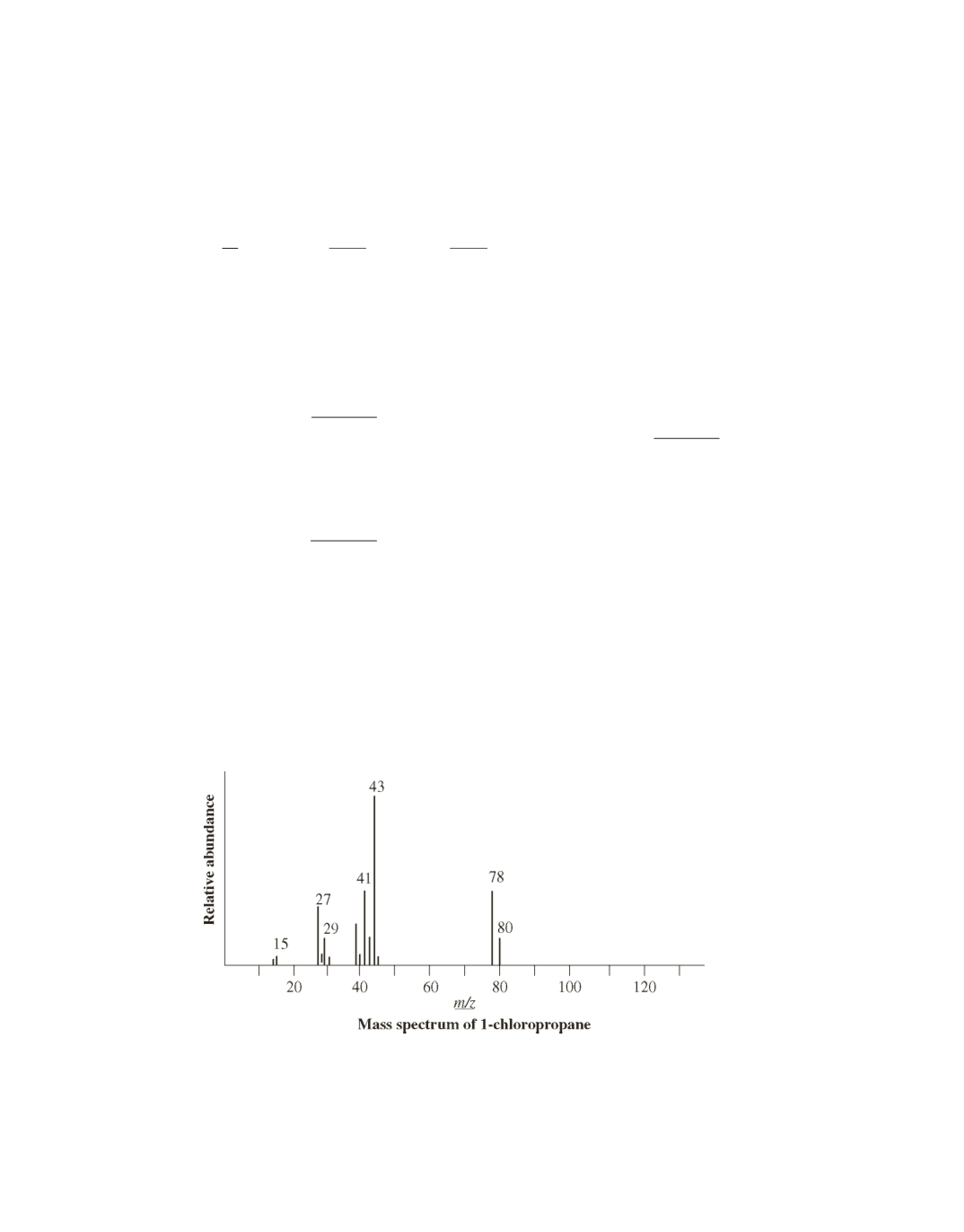
Because the compound contains chlorine, the M
+
2 peak is one-third the size of the M peak. Breaking
the weak C
¬
Cl bond heterolytically and, therefore, losing a chlorine atom from either the M
+
2 peak
(80
-
37) or the M peak (78
-
35) gives the base peak with
m
>
z
=
43
CH
3
CH
2
CH
2
([
]
+
.
).
















