
Chapter 13 435
Copyright © 2017 Pearson Education, Inc.
13.
The dominant fragmentation pathway in each case is loss of an alkyl radical via
a
-cleavage to form a
cation in which all the atoms have complete octets.
The base peak at
m
>
z
=
73 (88
-
15), due to loss of a methyl radical, indicates that
a.
is the mass
spectrum of
2-methoxy-2-methylpropane
.
The base peak at
m
>
z
=
59 (88
-
29), due to loss of an ethyl radical, indicates that
b.
is the mass
spectrum of
2-methoxybutane
.
The base peak at
m
>
z
=
45 (88
-
43), due to loss of a propyl radical, indicates that
c.
is the mass
spectrum of
1-methoxybutane
.
CH
3
CH
3
CCH
3
OCH
3
2-methoxy-2-methylpropane
CH
3
CH
2
CHCH
3
2-methoxybutane
1-methoxybutane
CH
3
CH
2
CH
2
CH
2
OCH
3
OCH
3
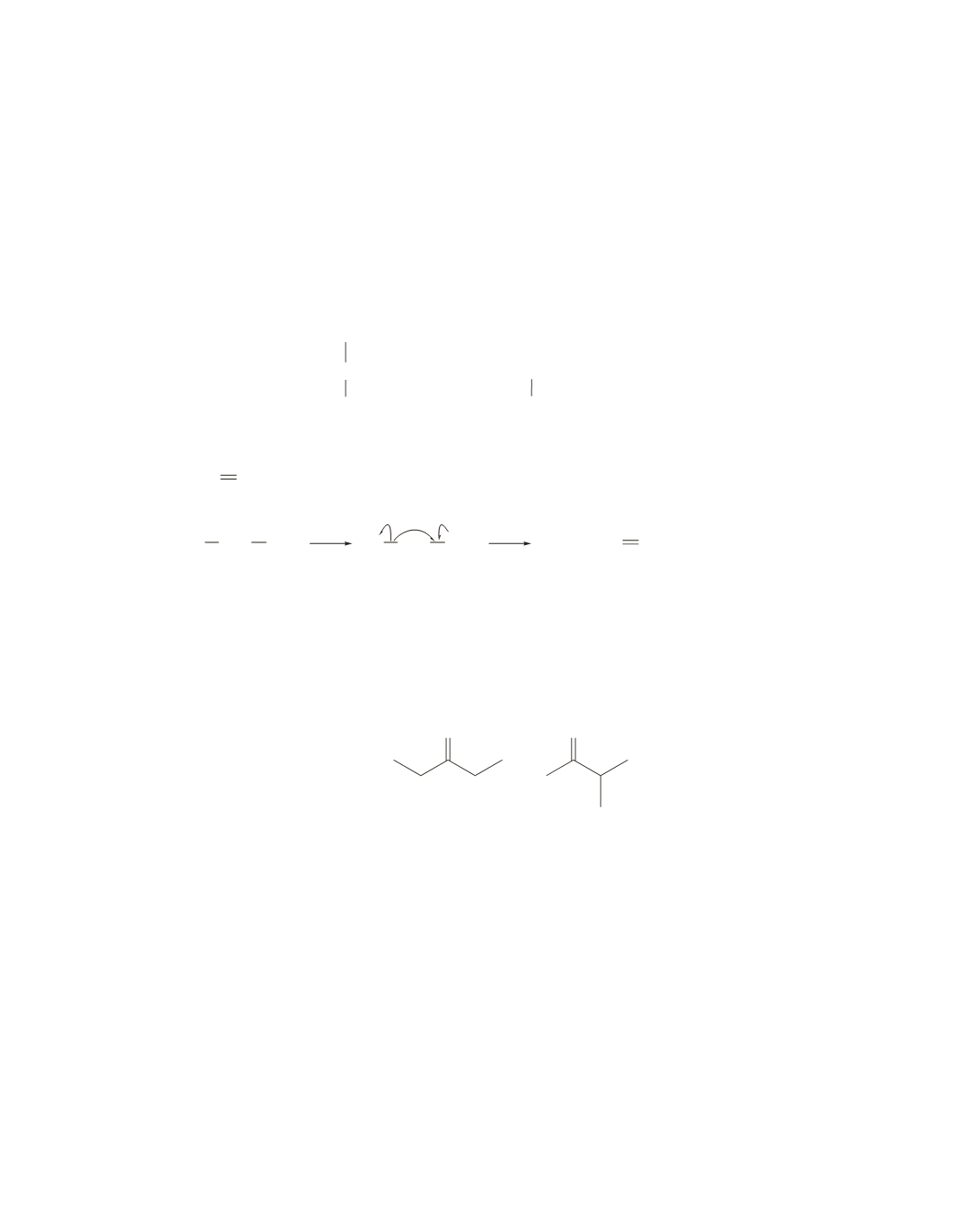
14.
R CH
2
OH
••
••
−
e
−
R CH
2
OH
•
••
+
R
•
+
CH
2
OH
+
••
CH
a.
b.
2
OH
+
m
/
z
=
31
15.
The molecular ions with
m
>
z
=
86 indicate that both ketones have the molecular formula C
5
H
10
O.
Spectrum
a.
shows a base peak at
m
>
z
=
43 for loss of a propyl (or isopropyl) radical (86
-
43),
indicating that it is the mass spectrum of either ketone A or ketone B because each of these has a propyl or
isopropyl group. The fact that the spectrum has a peak at
m
>
z
=
58, indicating loss of ethene (86
-
28),
indicates that the compound has a
g
-hydrogen that enables it to undergo a McLafferty rearrangement.
Therefore, the ketone must be
A
because
B
does not have a
g
-hydrogen.
O
A
O
B
Spectrum
b.
shows a base peak at
m
>
z
=
57 for loss of an ethyl radical (86
-
29), indicating that it is the
mass spectrum of A.
















