
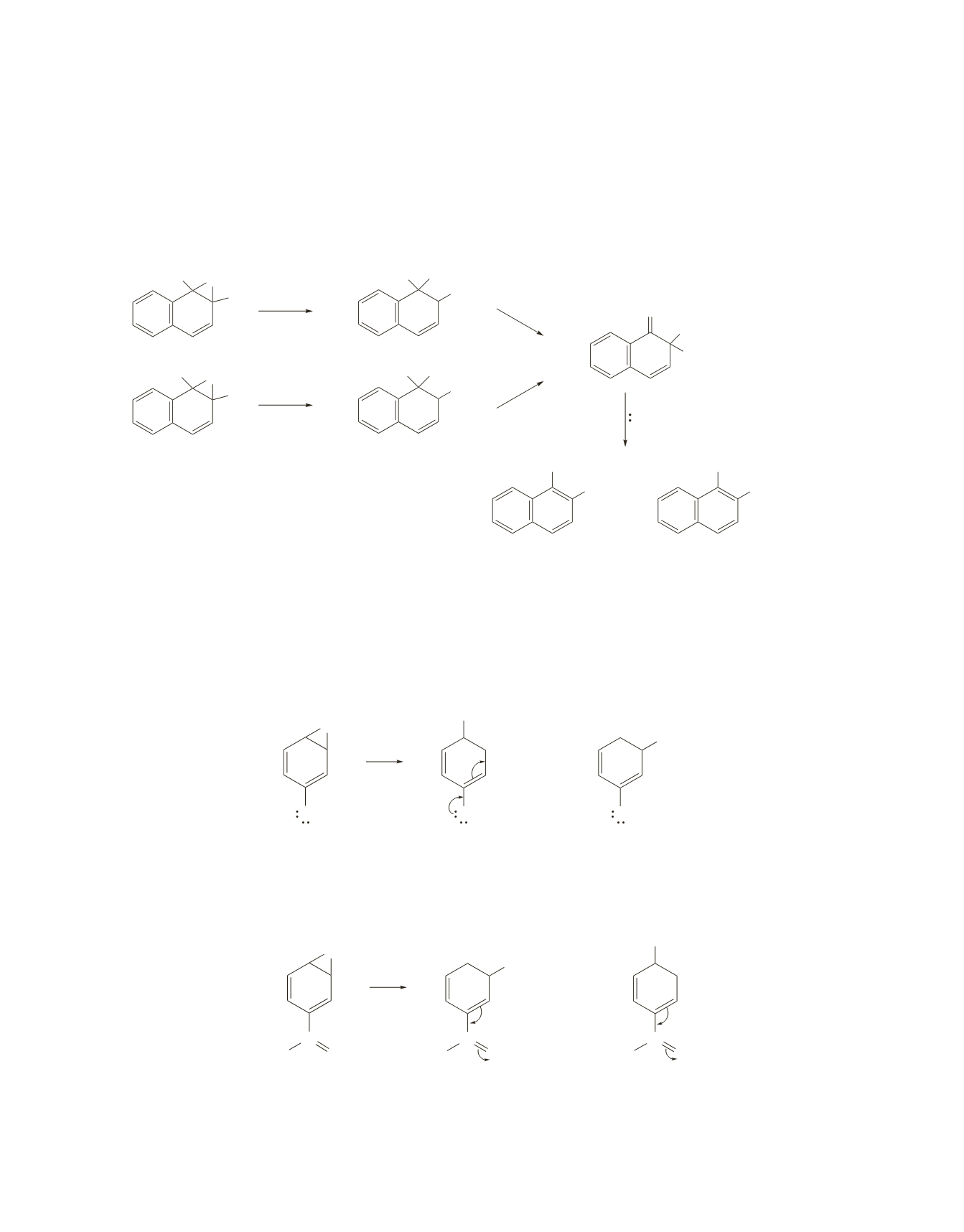
370 Chapter 10
Copyright © 2017 Pearson Education, Inc.
37.
The two arene oxides form the same major and minor products.
Each epoxide opens in the direction that forms the most stable carbocation. The carbocation undergoes an
NIH shift, and as a result of the NIH shift, both reactants form the same protonated ketone intermediate.
Because they form the same intermediate, they form the same products. The deuterium-containing product
is the major product, because in the last step of the reaction, it is easier to break a carbon–hydrogen bond
than a carbon–deuterium bond.
OH
D
HB
+
HB
+
BH
+
H OH
D
O D
H
+
D OH
H
+
+
OH
H
D
D
OH
H
OH
+
+
BD
+
NIH
shift
NIH
shift
+
major product
minor product
B
38.
Solved in the text.
39.
Each arene oxide opens in the direction that forms the more stable carbocation.
Therefore, the methoxy-substituted arene oxide opens so that the positive charge can be stabilized by elec-
tron donation from the methoxy group.
O
OCH
3
OH
OCH
3
OH
OCH
3
H
2
O
+
+
more stable
less stable
The nitro-substituted arene oxide opens in the direction that forms the carbocation intermediate that has its
positive charge farther away from the electron-withdrawing
NO
2
group.
O
N
N
N
H
2
O
+
+
more stable
less stable
OH
O O
O O
O O
+ +
−
−
−
+
OH
















