
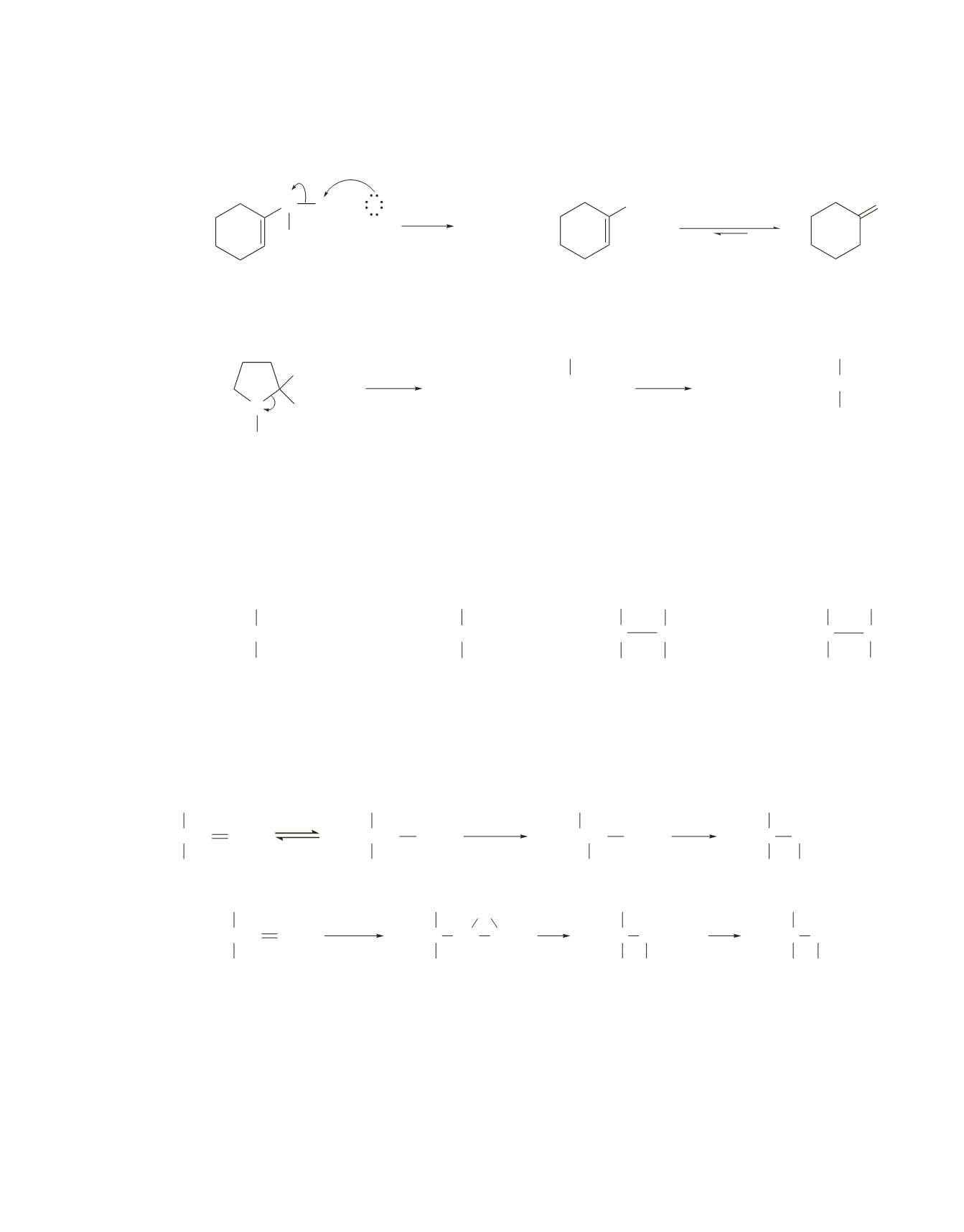
Chapter 10 365
Copyright © 2017 Pearson Education, Inc.
e.
Cleavage occurs by an
S
N
2
pathway because a primary carbocation or a vinylic cation is too unstable
to be formed.
CH
3
I
+
OH
tautomerization
O
enol
ketone
O
H
CH
3
+
I
−
f.
Cleavage occurs by an
S
N
1
pathway, because the tertiary carbocation is relatively stable.
HOCH
2
CH
2
CH
2
CCH
3
CH
3
O CH
3
CH
3
H
+
HOCH
2
CH
2
CH
2
CCH
3
CH
3
I
I
−
+
25.
Solved in the text.
26.
We saw that HCl does not cleave ethers because
Cl
-
is not a good enough nucleophile.
F
-
is an even poorer nucleophile, so HF cannot cleave ethers. Therefore, ethers can be cleaved only with
HBr or HI.
27.
a.
HOCH
2
CCH
3
OCH
3
CH
3
b.
CH
3
OCH
2
CCH
3
OH
CH
3
c.
CH
3
C CCH
3
OH OCH
3
CH
3
CH
3
d.
CH
3
C CCH
3
OCH
3
OH
CH
3
CH
3
28.
The reactivity of tetrahydrofuran is more similar to that of a noncyclic ether, because the five-membered
ring does not have the strain that makes the epoxide reactive.
29.
a.
+
+
CH
3
CH
3
CCH
CH
3
CH
2
CH
3
CH
3
CCH CH
3
CH
3
+
CH
3
CH
3
CCH CH
3
CH
3
H
2
SO
4
H
2
O
1,2-methyl
shift
OH CH
3
CH
3
C CHCH
3
H
+
CH
3
b.
CH
3
CH
3
CCH
CH
3
CH
2
CH
3
CH
3
C CH CH
2
CH
3
CH
3
O
−
CH
3
C CHCH
3
CH
3
H
−
HCl
MCPBA
CH
3
OH
CH
3
C CHCH
3
CH
3
O
















