
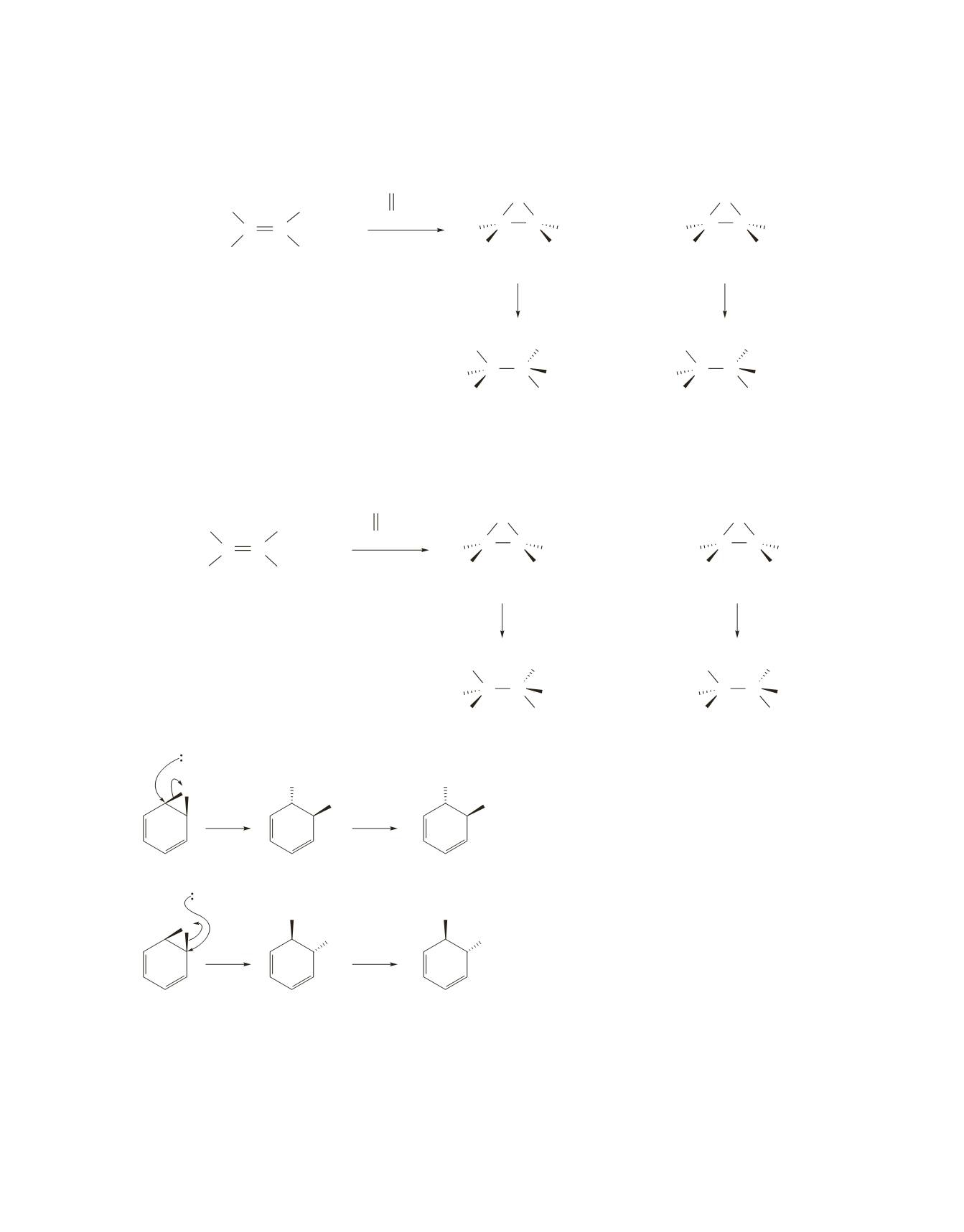
368 Chapter 10
Copyright © 2017 Pearson Education, Inc.
c.
Reaction of the cis alkene with a peroxyacid forms the two cis isomers. Hydroxide ion preferentially attacks
the least sterically hindered carbon of each epoxide. Therefore, the
S,S
and
R,R
products are formed.
H
C C OH
CH
2
CH
3
S S
H
H
CH
3
HO
H C C
CH
2
CH
3
R S
O
CH
3
H
H C C
CH
3
R S
O
CH
3
CH
2
H
+
+
HO
−
C C H
CH
3
R R
OH
H
HO
CH
3
CH
2
HO
−
H
CH
2
CH
3
O
RCOOH
CH
3
C C
d.
Reaction of the trans alkene with a peroxyacid forms the two trans isomers. Hydroxide ion preferen-
tially attacks the least sterically hindered carbon of each epoxide. Therefore, the
S,R
and
R,S
products
are formed.
CH
2
CH
3
C C OH
H
S
CH
2
CH
3
H
CH
3
HO
C C CH
3
OH
S R
R
H
H
CH
3
CH
2
HO
CH
2
CH
3
C C
H
R R
O
CH
3
H
H C C
CH
3
S S
O
H
CH
3
CH
2
+
+
HO
−
HO
−
H
H
O
RCOOH
CH
3
C C
34.
O
O
–
OH
Y
Y
Y
–
H – B +
B
+
O
O
–
Y
Y
–
OH
Y
H – B +
B
+
















