
372 Chapter 10
Copyright © 2017 Pearson Education, Inc.
c.
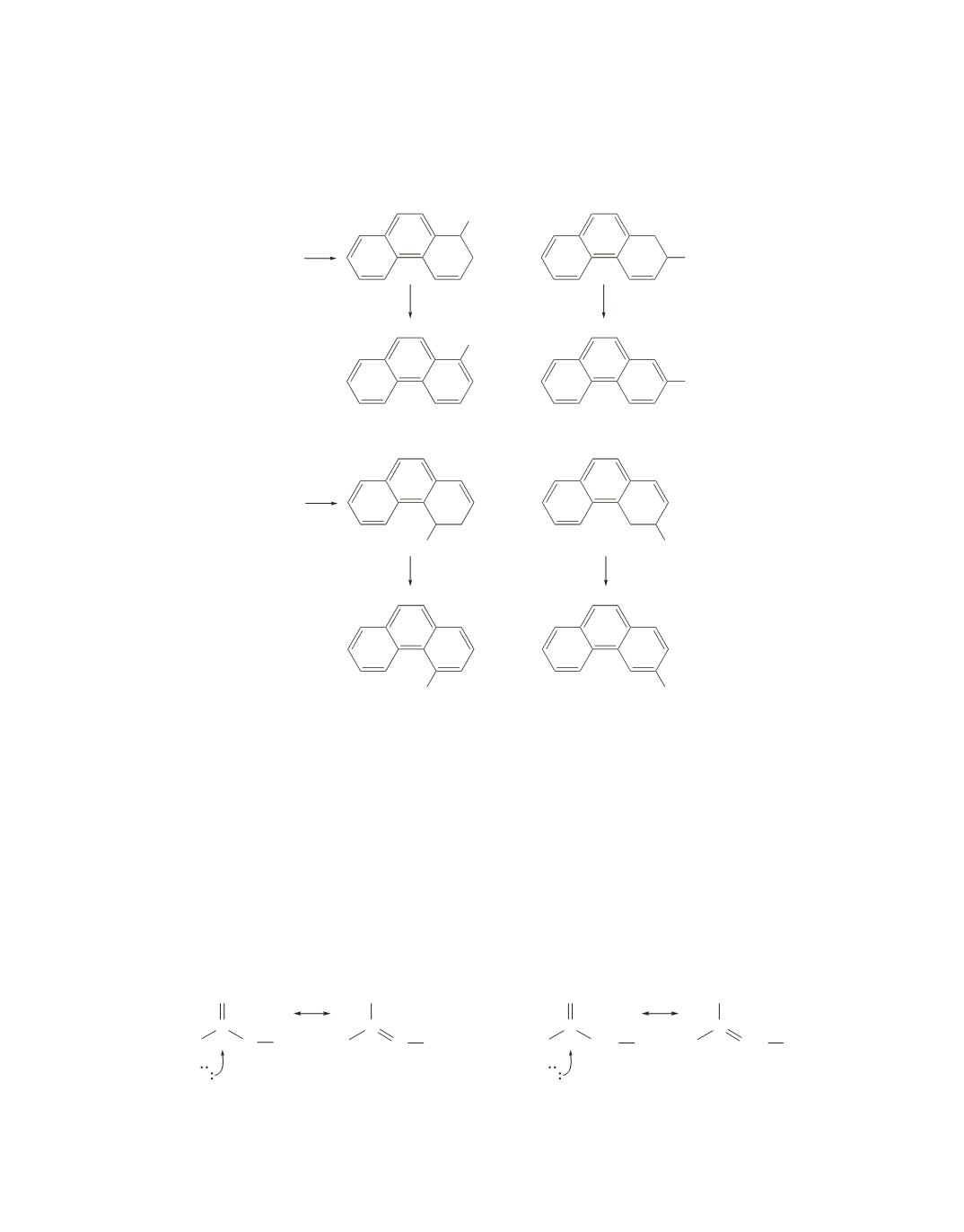
The two different carbocations formed by phenanthrene oxides II and III differ in stability. One
carbocation is more stable than the other because it can be stabilized by electron delocalization without
disrupting the aromaticity of the adjacent ring. The more stable carbocation leads to the major product.
II
OH
OH
+
OH
OH
+
major product
minor product
III
+
HO
OH
+
HO
OH
major product
minor product
+
+
+
+
d.
Phenanthrene oxide I is the most carcinogenic, because it is the only one that opens to form a carboca-
tion that cannot be stabilized without disrupting the aromaticity of the other ring(s).
42.
The drugs are metabolized as a result of reacting with water. Water adds to the electrophilic carbon of the
carbonyl group. The resonance contributor on the right (the one with separated charges) decreases the elec-
trophilicity of the carbonyl carbon, which makes it less reactive.
Xylocaine’s resonance contributor with separated charges is more stable than Novocaine’s resonance con-
tributor with separated charges because its positive charge is on nitrogen, which is a less electronegative
atom than oxygen. Because it is more stable, it makes a greater contribution to the hybrid. Therefore,
Xylocaine is less reactive and, consequently, has a longer half-life.
C
δ
−
O
O
C
O
−
O
δ
+
H
2
O
Novocaine
+
C
δ
−
O
NH
C
O
−
NH
H
2
O
Xylocaine
+
δ
+
















