
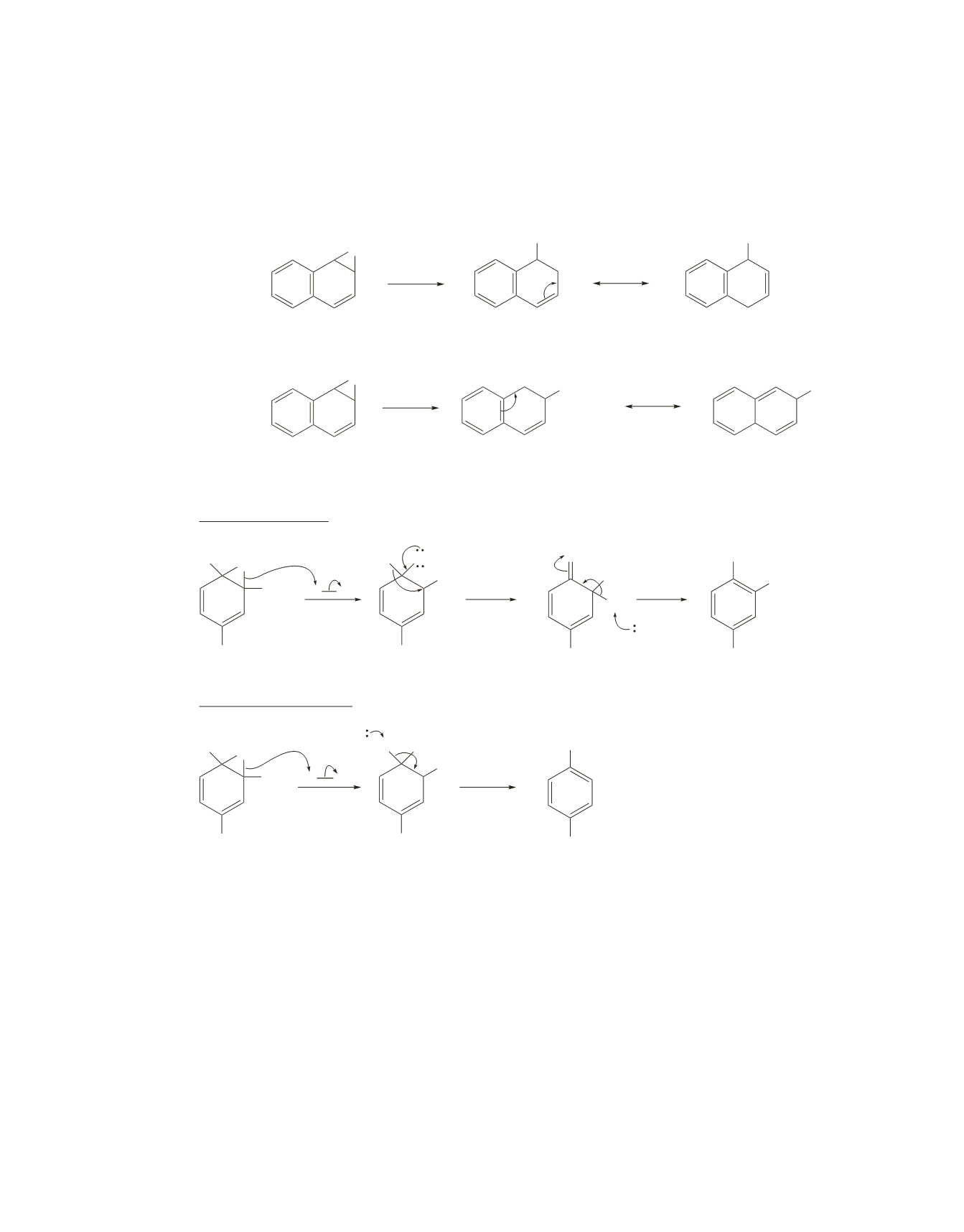
Chapter 10 369
Copyright © 2017 Pearson Education, Inc.
35.
The carbocation leading to 1-naphthol can be stabilized by electron delocalization without destroying the
aromaticity of the intact benzene ring. The carbocation leading to 2-naphthol can be stabilized by electron
delocalization only by destroying the aromaticity of the intact benzene ring. Therefore, the carbocation
leading to 1-naphthol is more stable.
O
OH
OH
+
+
carbocation that leads to 1-naphthol
O
+
OH
+
OH
carbocation that leads to 2-naphthol
36.
a. with an NIH shift
O D
CH
3
H
H B
OH D
H
OH
CH
3
CH
3
+
H
D
OH
CH
3
HB
+
+
+
+
NIH
shift
D
B
b. without an NIH shift
O D
CH
3
H
H B
+
OH D
H
OH
CH
3
CH
3
+
DB
+
+
B
















