
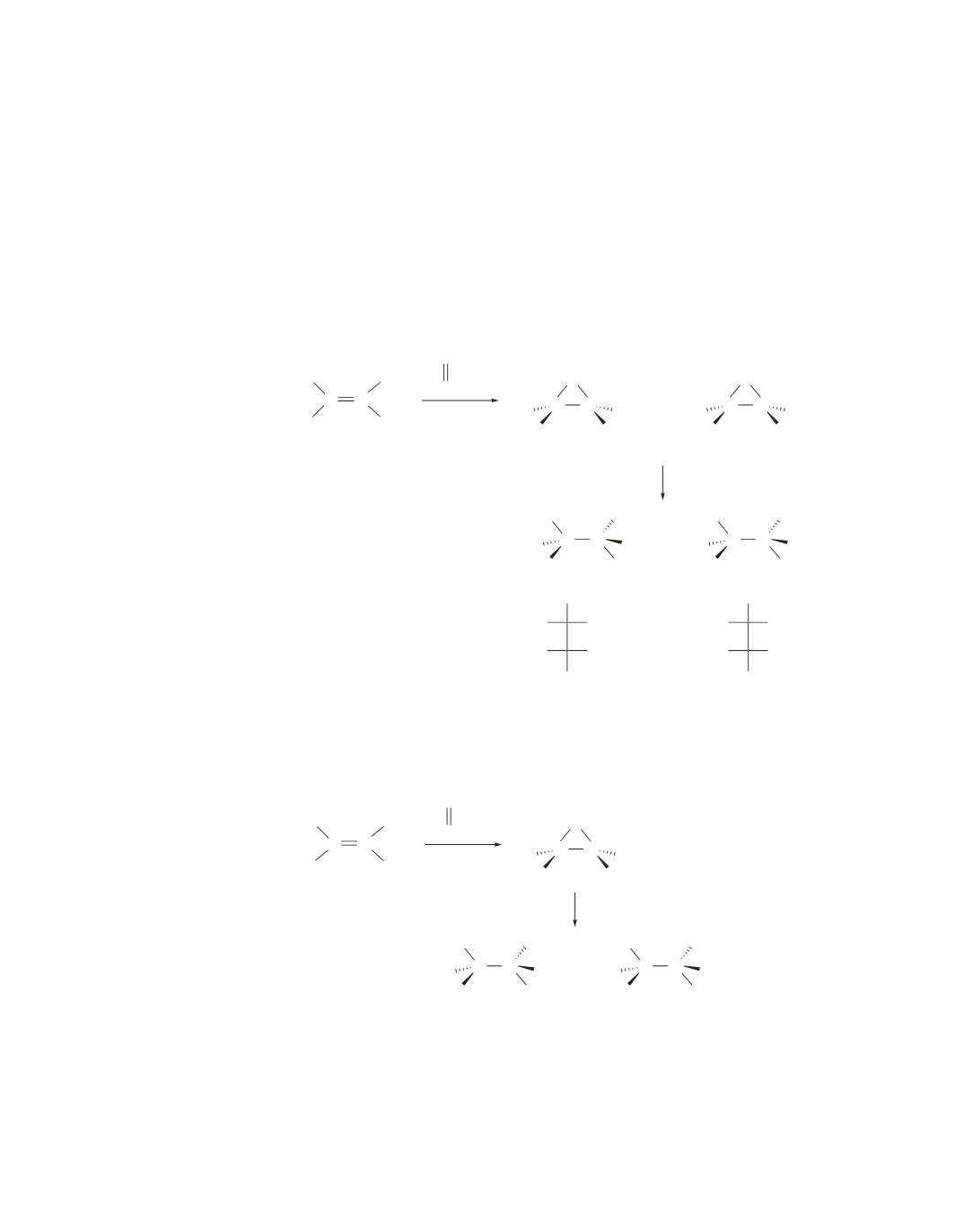
Chapter 10 367
Copyright © 2017 Pearson Education, Inc.
33.
a.
Reaction with a peroxyacid forms a product with two new asymmetric centers. Because it is a
syn addition reaction, the trans alkene forms the two trans products.
Hydroxide ion can attack either of the two asymmetric centers in the epoxide in an
S
N
2
reaction.
The asymmetric center that is attacked will have its configuration inverted.
The configuration of the other asymmetric center will not change.
Therefore, when hydroxide ion attacks the asymmetric center on the left of epoxide
A
, the
S,R
stereoisomer is formed; when it attacks the asymmetric center on the right of epoxide
A
, the
R,S
stereoisomer is formed. The
S,R
and
R,S
stereoisomers are identical; it is a meso compound.
Attack of hydroxide ion on epoxide
B
forms the same meso compound.
a meso compound
CH
3
HO
−
C C OH
H
S R
CH
3
H
CH
3
HO
C C CH
3
H
R S
OH
H
HO
CH
3
C C
H
R R
O
A
CH
3
H
H C C
CH
3
S S
O
B
H
CH
3
+
H
H
CH
3
O
RCOOH
CH
3
C C
=
CH
3
H OH
CH
3
H OH
R
S
CH
3
HO H
CH
3
HO H
S
R
b.
Reaction of the cis alkene with a peroxyacid forms only one peroxide (a meso compound). The meso com-
pound has the
R,S
configuration, so when it reacts with hydroxide ion, the
S,S
and
R,R
products are formed.
C C OH
CH
3
S S
H
H
HO
−
H
CH
3
HO
C C CH
3
H
R R
OH
H
HO
CH
3
C C
CH
3
R S
O
CH
3
H
H
CH
3
H
O
RCOOH
CH
3
C C
















