
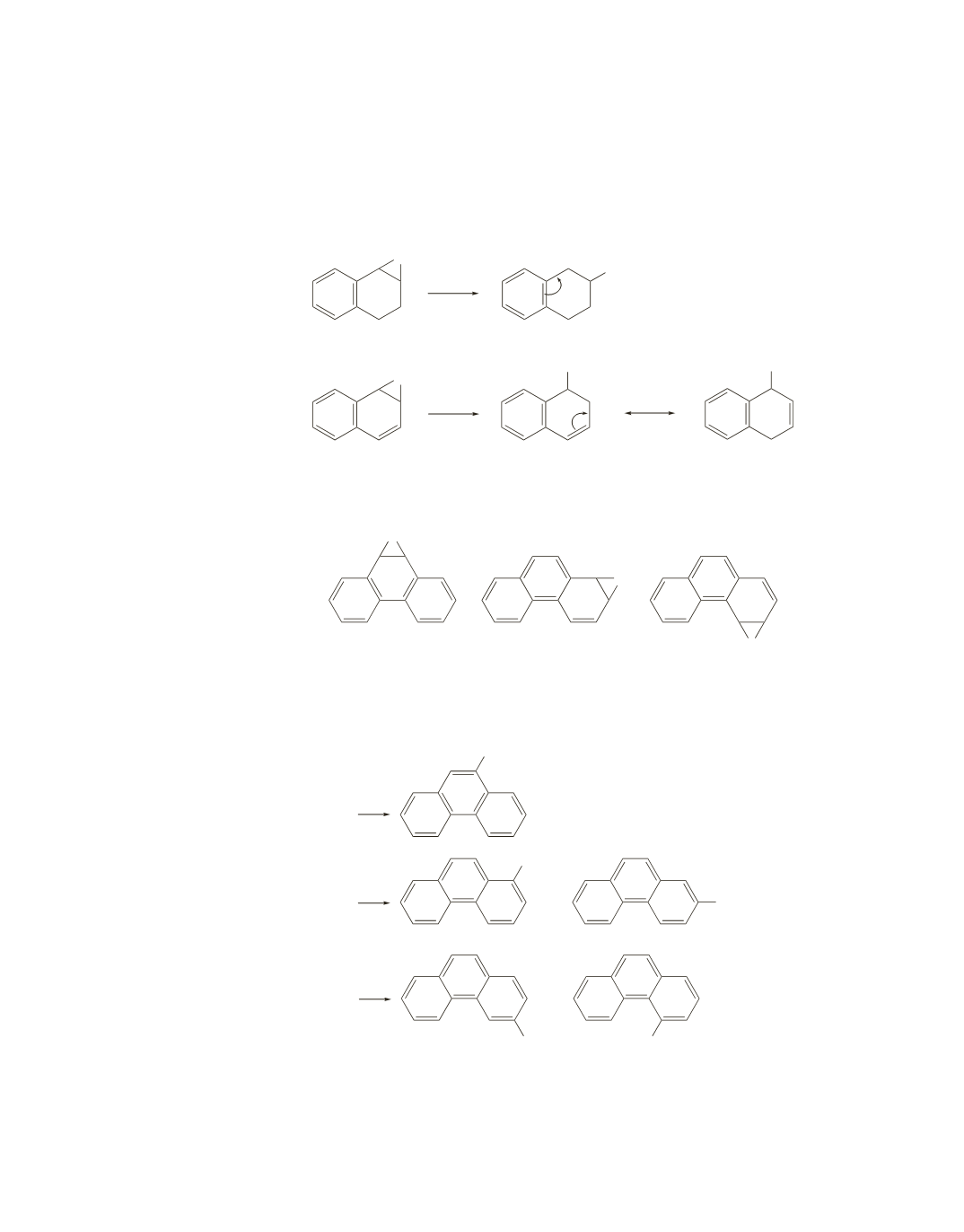
Chapter 10 371
Copyright © 2017 Pearson Education, Inc.
40.
The compound without the double bond in the second ring is more apt to be carcinogenic. It opens to
form a less stable carbocation than the other compound, because the carbocation can be stabilized by
electron delocalization only if the aromaticity of the benzene ring is destroyed. Because the carbocation
is less stable, it is formed more slowly, giving the carcinogenic pathway a better chance to compete with
ring-opening.
O
OH
O
+
+
+
less stable carbocation
more stable carbocation
OH
OH
41.
a.
Notice that a bond shared by two rings cannot be epoxidized.
O
O
O
I
II
III
b.
The epoxide ring in phenanthrene oxides II and III can open in two different directions to form two
different carbocations and, therefore, two different phenols.
I
II
OH
OH
OH
+
III
+
OH
HO
















