
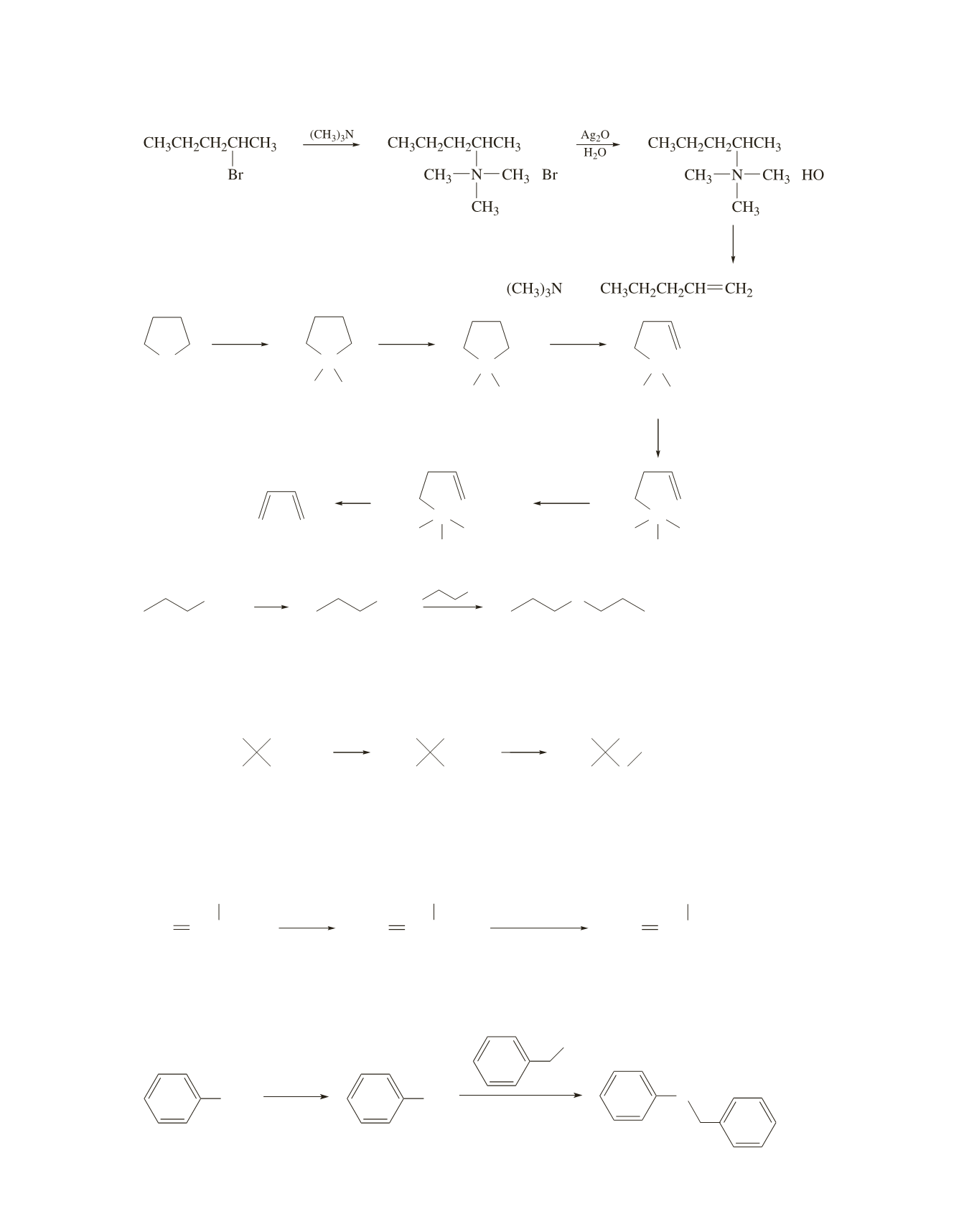
374 Chapter 10
Copyright © 2017 Pearson Education, Inc.
49.
a.
−
+
−
+
∆
+
b.
+
N
H
excess
CH3 I
K
2
CO
3
N
CH
3
CH
3
I
−
H
2
O
Ag
2
O
+
N
CH
3
CH
3
HO
−
∆
N
CH
3
CH
3
CH
3
I
N
+
CH
3
CH
3
CH
3
CH
3
H
2
O
Ag
2
O
I
−
N
+
CH
3
CH
3
HO
−
(CH
3
)
3
N +
50.
a.
+
SH
S
−
S
Br
Br
−
+
H
2
O
HO
−
b.
The synthesis must be done with
tert
-butylthiol and a methyl halide. It cannot be done with methane-
thiol and
tert-
butyl bromide because a tertiary alkyl halide cannot undergo an
S
N
2
reaction. Therefore,
it would form an elimination product rather than a substitution product.
+
Br
SH
HO
−
−
−
CH
3
Br
+
H
2
O
S
S
c.
The highest yield of the target molecule is obtained by having the less substituted of the two R groups
of the thioether be the alkyl halide and the more substituted be the thiol.
CH
2
CHCHSH
CH
3
HO
−
CH
2
CHCHS
−
CH
3
CH
3
CH
2
Br
CH
2
CHCHSCH
2
CH
3
CH
3
+
Br
−
+
H
2
O
d.
The synthesis must be done with these reagents because the
sp
2
carbon of the benzene ring cannot un-
dergo back-side attack.
HO
−
SH
Br
S
−
S
+
Br
−
+
H
2
O
















