
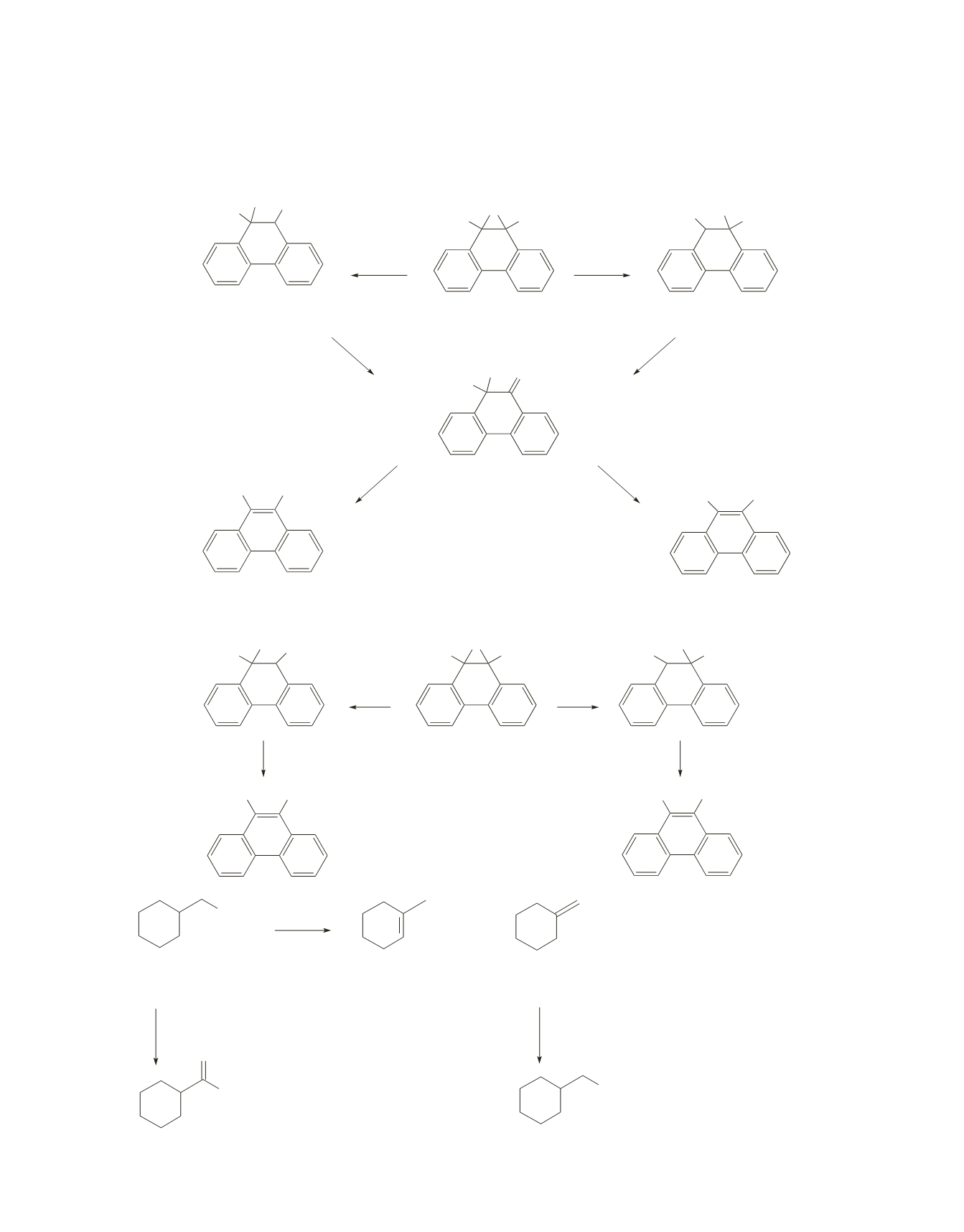
380 Chapter 10
Copyright © 2017 Pearson Education, Inc.
65.
a.
If an NIH shift occurs, both carbocations will form the same intermediate ketone. Because it is about
four times easier to break a C—H bond
1
k
3
2
compared with a C—D bond
1
k
4
2
,
about 80
,
of the
deuterium will be retained.
D OH
H
+
O
D H
k
1
H
2
O
k
2
H
2
O
HO
D
+
H
OD
NIH
shift
NIH
shift
k
4
k
3
OH D
OH
H
H
b.
If an NIH shift does not occur, 50
,
of the deuterium will be retained, because the epoxide can open
equally easily in either direction
1
k
1
is equal to
k
2
2
and subsequent loss of
H
+
or
D
+
is fast.
D H
+
H
H
+
O
HO
H
2
O
k
1
H
2
O
k
2
OH
D
D HO
OH H
D
+
H
+
+
D
+
66.
OH
unknown
alcohol
H
O
+
NaOCl
H
2
SO
4
major
product
minor
product
OH
1.
2.
R
2
BH/THF
H
2
O
2
, HO
−
, H
2
O
original unknown
alcohol
CH
3
COOH
0 ˚C
















