
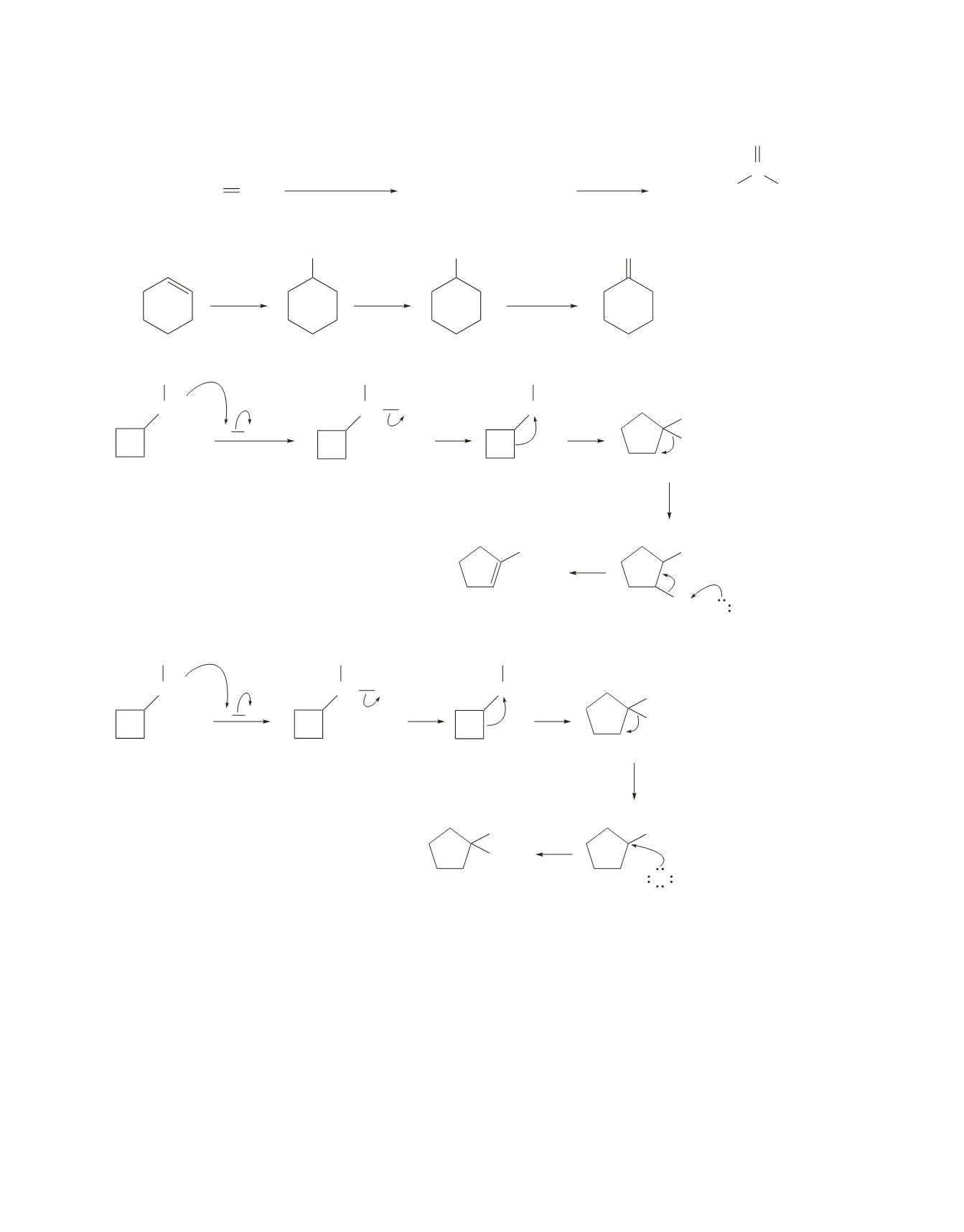
382 Chapter 10
Copyright © 2017 Pearson Education, Inc.
b.
CH
3
CH
2
CH
2
CH
3
CH
2
CH CH
2
1. R
2
BH/THF
2. H
2
O
2
, HO – , H
2
O
NaOCl
CH
3
COOH
0
°
C
CH
3
CH
2
CH
2
CH
2
OH
O
C
H
c.
HBr
HO
−
Br
OH
O
NaOCl
CH
3
COOH
0 ºC
70.
CHOH
OSO
3
H
H
+
+
• •
• •
CH
3
CH
CH
3
CH
CH
3
OH
H
H
+
+
+
CH
3
CH
3
CH
3
H
+
H
2
O
H
2
O
H
3
O
+
71.
CHOH
Br
H
+
+
• •
• •
CH
3
CH
CH
3
CH
CH
3
OH
H
+
Br
Br
−
CH
3
CH
3
H
CH
3
+
72.
Diethyl ether
is the ether that would be obtained in greatest yield, because it is a symmetrical ether.
Because it is symmetrical, only one alcohol is used in its synthesis, so only one ether is formed.
In contrast, the synthesis of an unsymmetrical ether requires two different alcohols, so three different ethers
are formed.
















