
384 Chapter 10
Copyright © 2017 Pearson Education, Inc.
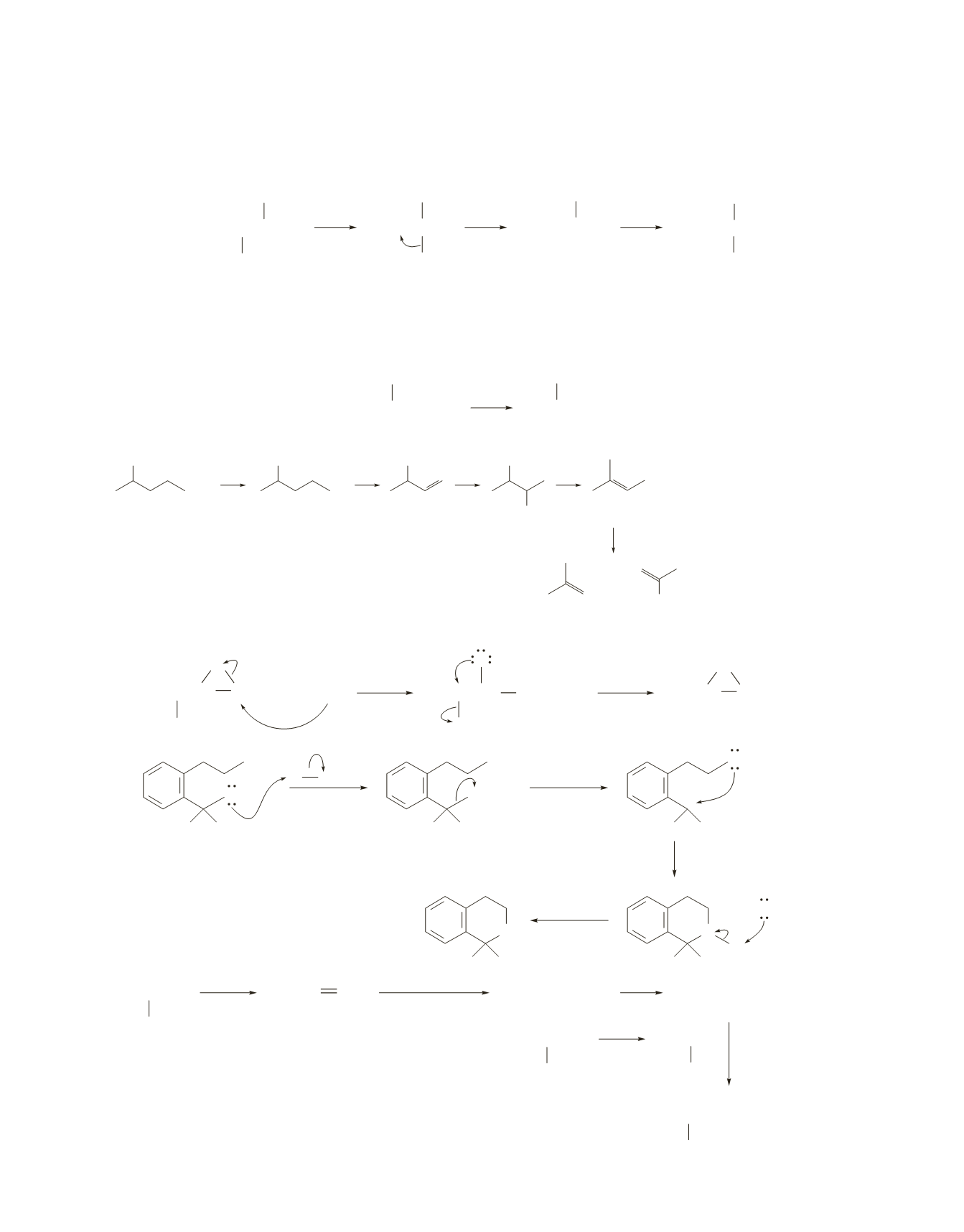
75.
3-Methyl-2-butanol is a secondary alcohol and, therefore, undergoes an
S
N
1
reaction. The secondary
carbocation intermediate rearranges to a more stable tertiary carbocation.
CH
3
CHCHCH
3
OH
CH
3
CH
3
CHCCH
H
3
CH
3
+
CH
3
CH
2
CCH
3
CH
3
+
CH
3
CH
2
CCH
3
CH
3
HBr
Br
3-methyl-2-butanol
2-Methyl-1-propanol is a primary alcohol and, therefore, undergoes an
S
N
2
reaction. Because carbocations
are not formed in
S
N
2
reactions, a carbocation rearrangement cannot occur.
CH
3
CHCH
2
OH
CH
3
CHCH
2
Br
HBr
2-methyl-1-propanol
CH
3
CH
3
76.
OH
Cl
OH
H
O
O
+
C
D
E
F
B
A
77.
a.
CH
3
CHCH CH
2
O
O
Cl
CH
3
CH CHCH
2
OCH
3
+
CH
3
O
−
• •
• •
•
•
CH
3
CHCH
Cl
CH
2
OCH
3
O
+
Cl
−
−
b.
OH
+
H OSO
3
H
OH
OH
OH
OH
H
+
H
H
2
O
+
+
H
3
O
+
O
O
78.
PBr
3
CH
3
CH CH
2
CH
3
CHOH
CH
3
H
2
SO
4
1. R
2
BH/THF
2. H
2
O
2
, HO
−
, H
2
O
CH
3
CH
2
CH
2
OH
CH
3
CH
2
CH
2
Br
CH
3
CHOCH
2
CH
2
CH
3
CH
3
CH
3
CHOH
CH
3
NaH
CH
3
CHO
−
CH
3
pyridine
















