
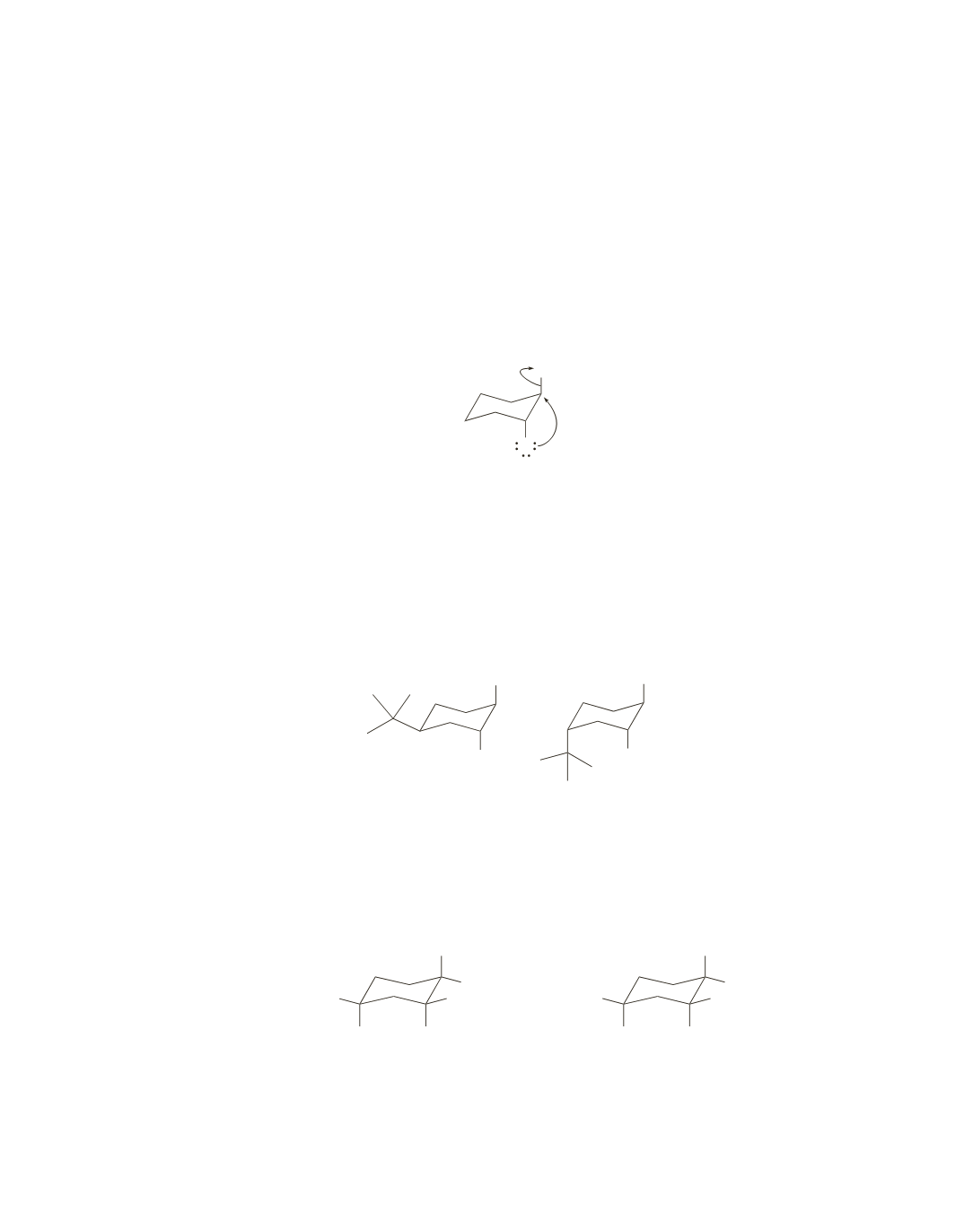
Chapter 10 389
Copyright © 2017 Pearson Education, Inc.
The second reaction is also an
S
N
2
reaction and is fast because the strain of the three-membered ring
and the positive charge on the nitrogen make the amine a very good leaving group.
b.
The HO group is bonded to a different carbon because
HO
-
attacks the least sterically hindered carbon
of the three-membered ring.
90.
B
is the fastest reaction;
A
is the slowest reaction.
To form the epoxide, the alkoxide ion must attack the back side of the carbon that is bonded to Br. This
means that the OH and Br substituents must both be in axial positions. To be 1,2-diaxial, they must be
trans to each other.
Br
O
−
A
does not form an epoxide, because the OH and Br substituents are cis to each other.
B
and
C
can form epoxides because the OH and Br substituents are trans to each other.
The rate of formation of the epoxide is given by
k K
eq
,
where
k
is the rate constant for the substitution
reaction and
K
eq
is the equilibrium constant for the [equatorial]
>
[axial] conformers.
When the OH and Br substituents are in the required diaxial position, the large
tert
-butyl substituent is in
the equatorial position in
B
and in the axial position in
C
.
Br
OH
Br
OH
C
B
Because the more stable conformer has the large
tert
-butyl group in the equatorial position, the OH and
Br substituents are in the required diaxial position in the more stable conformer of
B
1
K
eq
is large
2
,
whereas the OH and Br substituents are in the required diaxial position in the less stable conformer of
C
1
K
eq
is small
2
. Therefore,
B
reacts faster than
C
.
Br
H
H
OH
C(CH
3
)
3
H
Br
H
H
OH
H
(CH
3
)
3
C
B
C
















