
392 Chapter 10
Copyright © 2017 Pearson Education, Inc.
96.
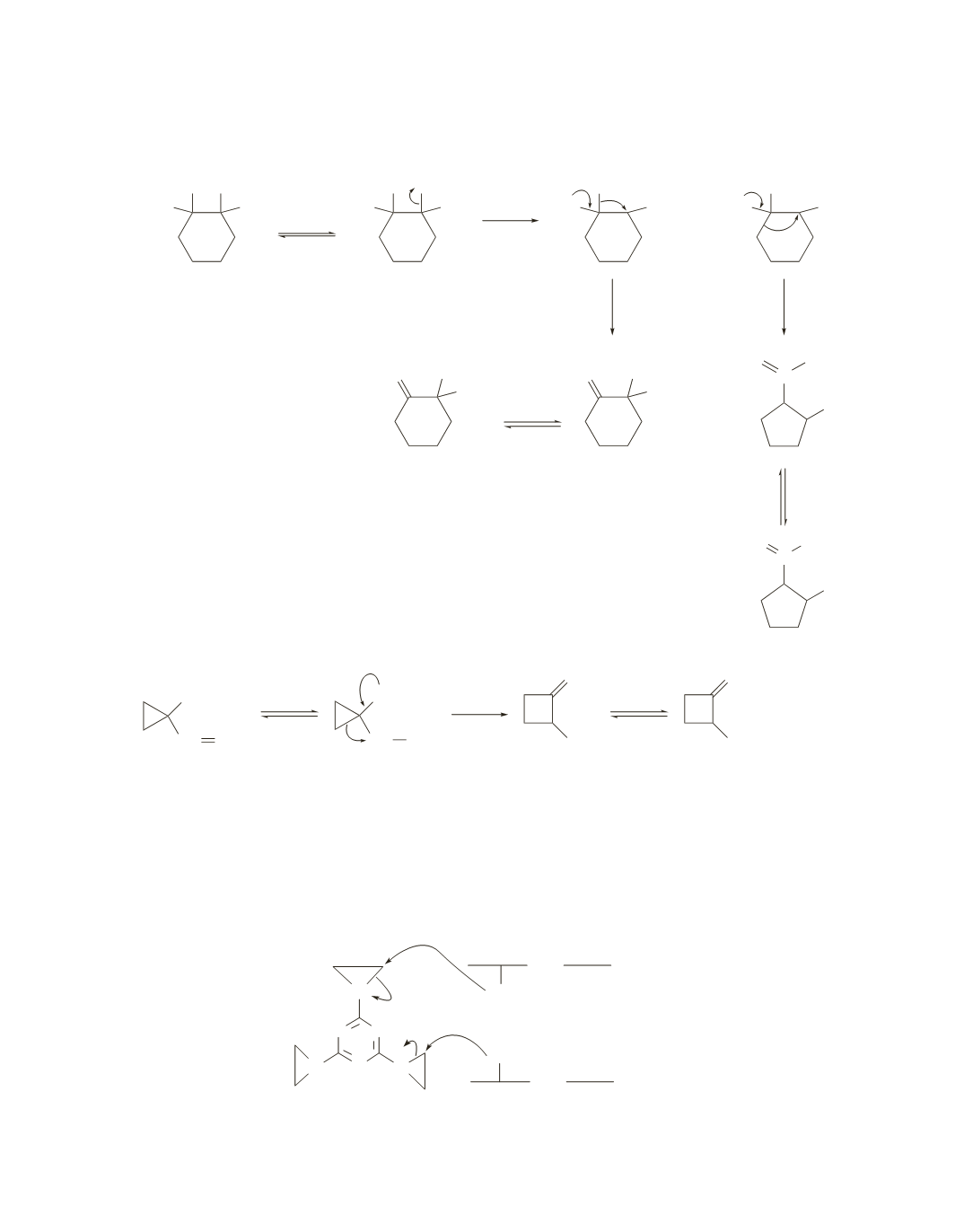
Notice that the initially generated carbocation can undergo either a 1,2-methyl shift or a 1,2-alkyl shift.
a.
HO OH
CH
3
CH
3
HO OH
CH
3
CH
3
H
+
CH
3
CH
3
HO
+
or
CH
3
CH
3
HO
+
• •
H
2
SO
4
C
CH
3
HO
CH
3
CH
3
HO
CH
3
CH
3
O
+
+
+
H
3
O
+
C
CH
3
O
+
+
H
3
O
+
• •
• •
• •
CH
3
CH
3
b.
H
2
SO
4
CH
3
OH
H
3
O
+
+
+
OH
CH CH
2
OH
CH CH
3
+
• •
• •
CH
3
O
97.
a.
A nitrogen is a stronger base than an oxygen, so unlike an epoxide that can be opened without the
oxygen being protonated, the three-membered nitrogen-containing ring has to be protonated to improve
the leaving propensity of the group.
b.
A nucleophile such as an
NH
2
group on a chain of DNA can react with the three-membered ring. If
a nucleophile on another chain of DNA reacts with another of the three-membered rings in this com-
pound, the two DNA chains will be cross-linked.
+
..
N
N
N
N
N
NH
DNA
NH
2
DNA
NH
2
H
+
..
















