
396 Chapter 11
Copyright © 2017 Pearson Education, Inc.
Solutions to Problems
1.
All the reactions occur because in each case, the reactant acid is a stronger acid than the product acid
(methane, p
K
a
60).
2.
The greater the polarity of the carbon–metal bond, the more reactive the organometallic compound.
Polarity depends on the difference in electronegativity between the atoms forming the bond; the greater the
difference in electronegativity, the more polar the bond.
C Li
C Na
2.5
−
1.0 = 1.5
2.5
−
0.9 = 1.6
The electronegativity difference between carbon and sodium is greater than that between carbon and lith-
ium. Therefore, organosodium compounds (with a more polar carbon–metal bond) are more reactive than
organolithium compounds.
3.
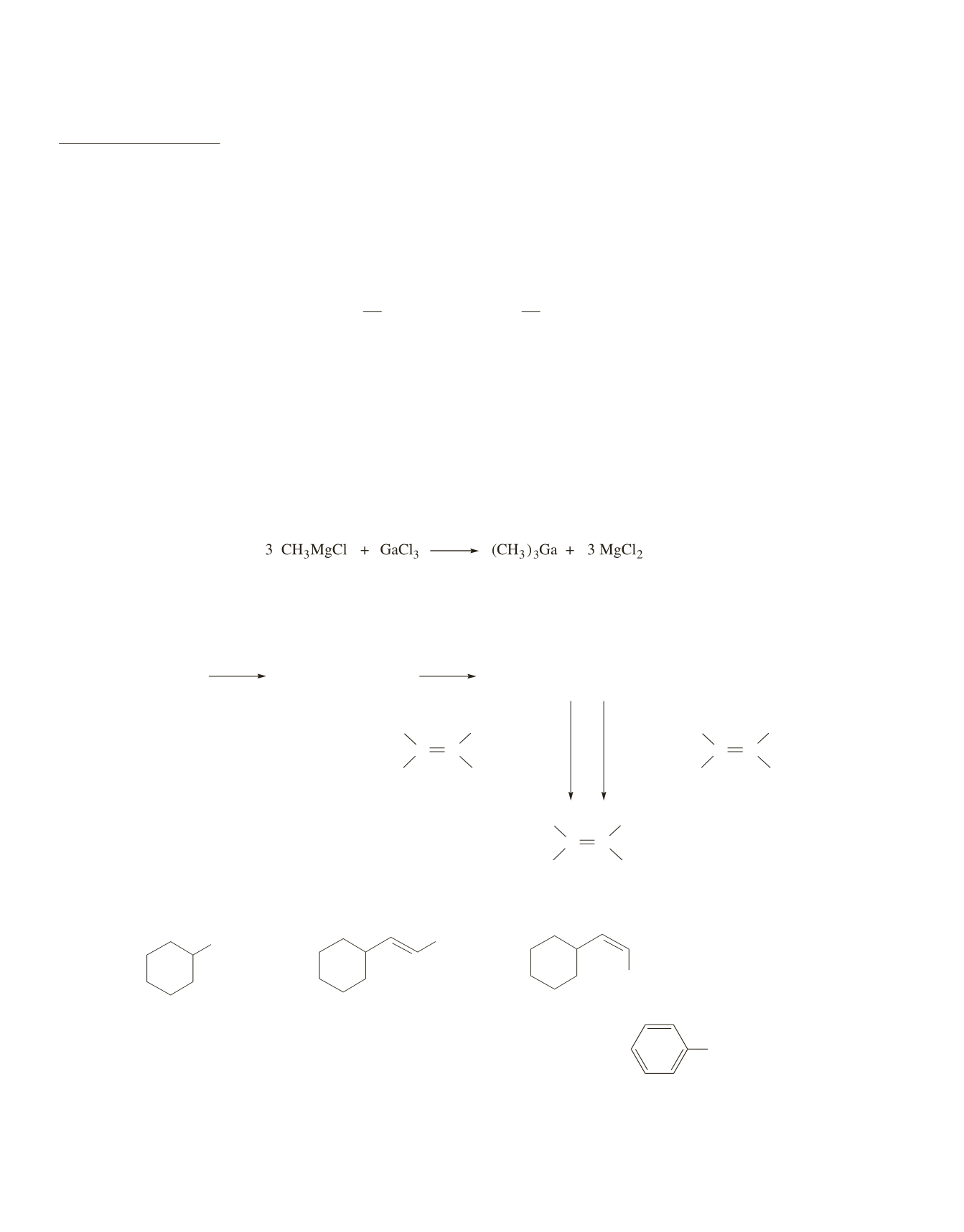
Transmetallation will occur if the new metal’s electronegativity is closer to that of carbon. Because gallium
(1.8) is more electronegative than magnesium (1.2) and, therefore, closer to the electronegativity of carbon,
transmetallation occurs. Notice that after transmetallation, the number of alkyl groups attached to gallium
is the same as the number of chorines that were attached to it in the reactant.
4.
Solved in the text.
5.
The Br is replaced by the alkyl group of the organocuprate.
2 CH
3
(CH
2
)
3
CH
2
Br
2 CH
3
(CH
2
)
3
CH
2
Li
(CH
3
CH
2
CH
2
CH
2
CH
2 2
) CuLi
2 Li
hexane
CuI
THF
C C
H
(CH
2
)
12
CH
3
H
CH
3
(CH
2
)
7
C C
H
(CH
2
)
7
CH
2
Br
H
CH
3
(CH
2
)
7
C C
H
(CH
2
)
12
CH
3
H
or
BrCH
2
(CH
2
)
2
6.
Solved in the text.
7.
Br
a.
Br
b.
Br
c.
8.
a.
CH
3
CH
2
CH
2
CH
2
CH
2
OH
b.
CH
3
CH
“
CHCH
2
CH
2
OH
c.
CH
2
CH
2
CH
2
OH
















