
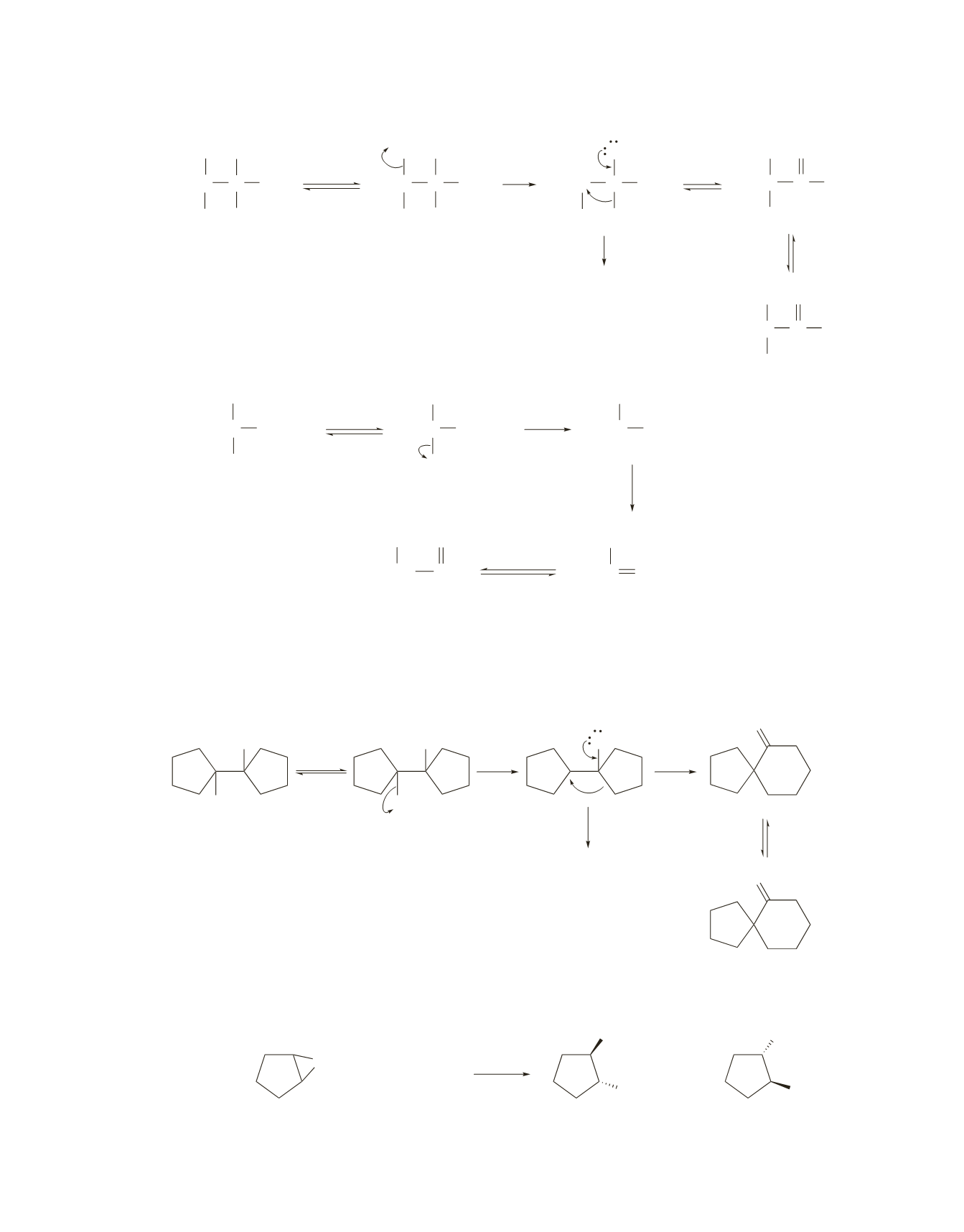
Chapter 10 391
Copyright © 2017 Pearson Education, Inc.
92.
CH
3
C
OH
CH
3
C
OH
CH
3
CH
3
H
2
SO
4
CH
3
C
OH
CH
3
C
OH
CH
3
CH
3
CH
3
C
CH
3
C
OH
CH
3
CH
3
H
+
+
+
H
2
O
CH
3
C
CH
3
C CH
3
CH
3
C
CH
3
CH
3
C
O
CH
3
+
H
3
O
+
+
CH
3
OH
93.
a.
CH
3
C
CH
3
OH
CH
2
OH
H
2
SO
keto-enol
interconversion
4
H
+
+
H
2
O
H
3
O
+
+
CH
3
C
CH
3
OH
CH
2
OH
CH
3
C
CH
3
CH
2
OH
+
CH
3
CH
CH
3
CH
CH
3
C
CH
3
CHOH
O
b.
Dehydration of the primary alcohol group cannot occur, because it cannot lose water via an E1 path-
way, since a primary carbocation cannot be formed. It cannot lose water via an E2 pathway because the
b
-carbon is not bonded to a hydrogen. However, dehydration of the tertiary alcohol group can occur.
The product is an enol that tautomerizes to an aldehyde.
94.
OH
OH
OH
OH
OH
H
2
SO
4
H
+
H
3
O
+
+
+
+
H
2
O
+
OH
O
95.
Cyclopentene oxide undergoes back-side attack by the nucleophile, so the two substituents in the products
are trans to each other. Therefore, both the
R,R
-isomer and the
S,S
-isomer are formed.
O
cyclopentene
oxide
+
CH
3
NHCH
3
R
,
R
-isomer
N(CH
3
)
2
OH
+
S
,
S
-isomer
OH
N(CH
3
)
2
dimethylamine
















