
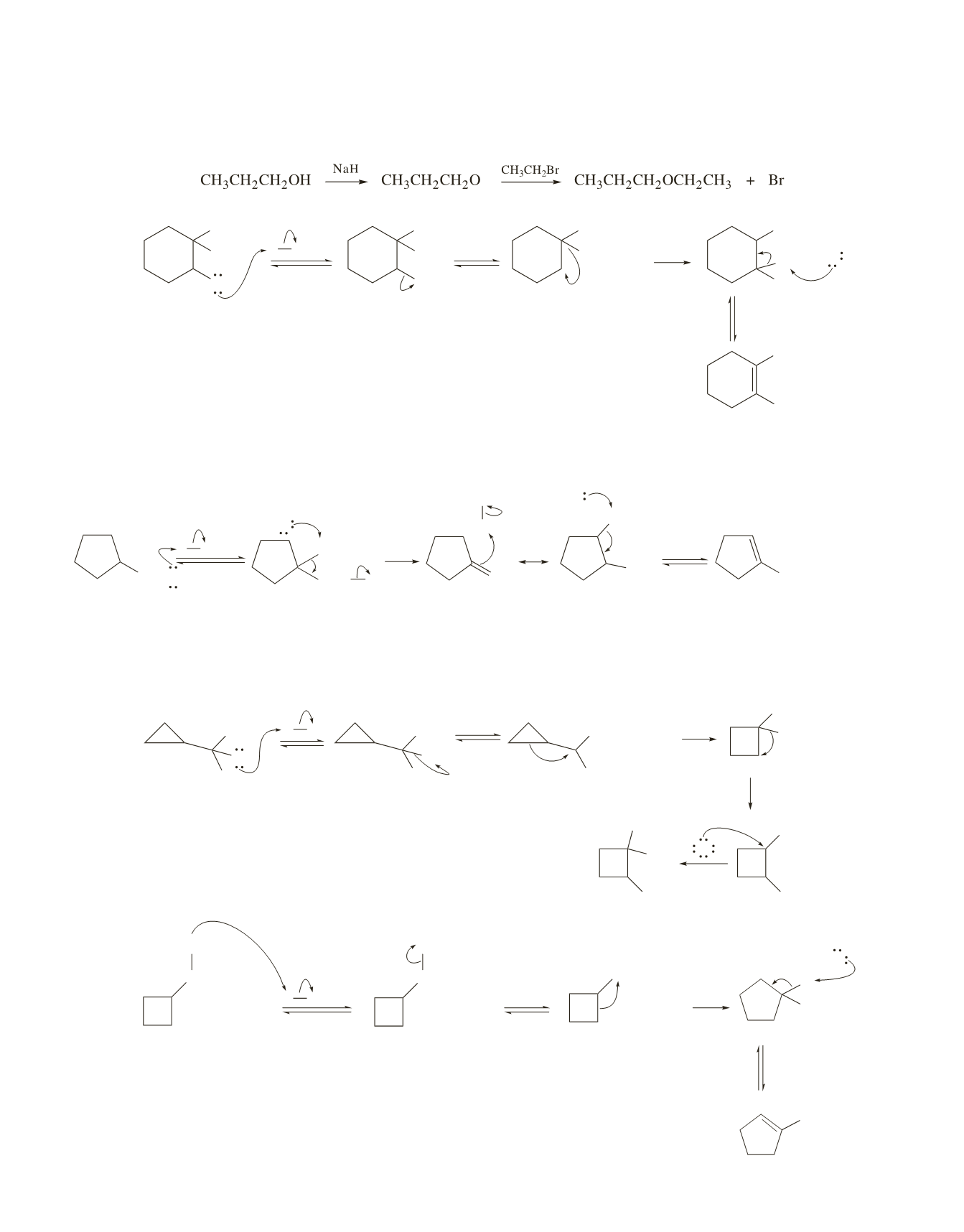
362 Chapter 10
Copyright © 2017 Pearson Education, Inc.
b.
It could be synthesized by a Williamson ether synthesis. (See Section 9.15 on page 443 of the text.)
−
−
16.
a.
OH
CH
3
CH
3
OSO
3
H
H
O
H
CH
3
CH
3
+
H
CH
3
CH
3
+
H
2
O
+
CH
3
CH
3
+
H H
2
O
CH
3
CH
3
+
H
3
O
+
b.
Because a primary carbocation cannot be formed, the dehydration is an E2 reaction. The alkene that results
is protonated, and the proton that is removed results in formation of the most stable alkene.
CH
2
OH
CH
2
H
O
E2
H
H
+
H
2
O
CH
2
HOH
H
+
H
2
O
+
H
2
O
CH
3
H
CH
3
+
H
3
O
+
+
OSO
3
H
H
17.
The strained three-membered ring causes a tertiary carbocation with a three-membered ring to be less
stable than a secondary carbocation with a less strained four-membered ring. The secondary carbocation
then rearranges to a tertiary carbocation.
a.
OH
OH
CH
3
H
CH
3
CH
3
+
CH
3
CH
3
CH
3
+
H
2
O
+
CH
3
CH
3
+
CH
3
+
CH
3
CH
3
Br
4
3
2
1
3
2 1
4
−
Br
−
Br
CH
3
Br
H
b.
CHCH
2
CH
3
+
+
CHCH
2
CH
3
CHCH
2
CH
3
OH
H
+
CH
2
CH
3
H
2
O
H
CH
2
CH
3
OH
• •
•
•
OSO
3
H
H
−
+
H
3
O
+
















