
Chapter 8 271
Copyright © 2017 Pearson Education, Inc.
23.
When
para
-nitrophenol loses a proton, the negative charge in the conjugate base can be delocalized onto
the nitro substituent. Therefore, the
para
–nitro substituent decreases the p
K
a
both by resonance electron
withdrawal and by inductive electron withdrawal.
OH
N
−
O O
O
N
−
O O
+
H
+
+
+
−
When
meta
-nitrophenol loses a proton, the negative charge in the conjugate base cannot be delocalized
onto the nitro substituent. Therefore, the
meta
-nitro substituent can decrease the p
K
a
only by inductive
electron withdrawal. Therefore, the para isomer has a lower p
K
a
.
OH
N
O
−
O
O
N
O
−
O
+
+
+
H
+
−
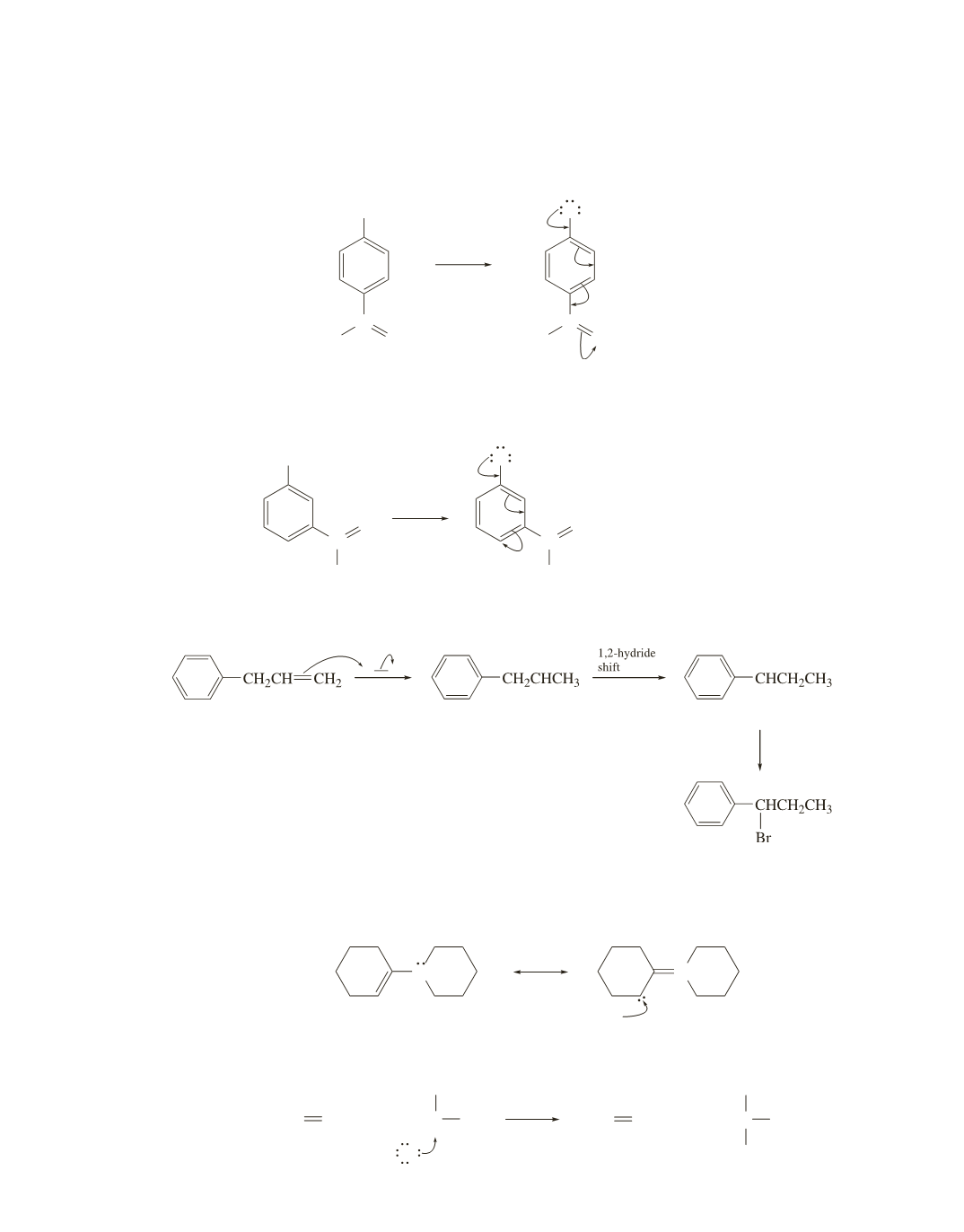
24.
Recall that if a more stable carbocation can be formed as a result of carbocation rearrngement, rearrange-
ment will occur.
H Br
Br
−
secondary carbocation
secondary benzylic cation
+
+
25.
Solved in the text.
26.
The resonance contributors show that one of the atoms has a partial negative charge. Therefore, that is the
atom that is more apt to be protonated.
N
N
−
+
site of protonation
27.
a.
The more reactive double bond is the one that forms a tertiary carbocation.
CH
2
CH
3
Br
CHCH
2
CH
2
CH
2
C CH
3
CH
2
CH
3
Br
+
−
CHCH
2
CH
2
CH
2
C CH
3
















