
268 Chapter 8
Copyright © 2017 Pearson Education, Inc.
8.
−
O O
−
C
O
H O
−
C
O
H
C
O
OH
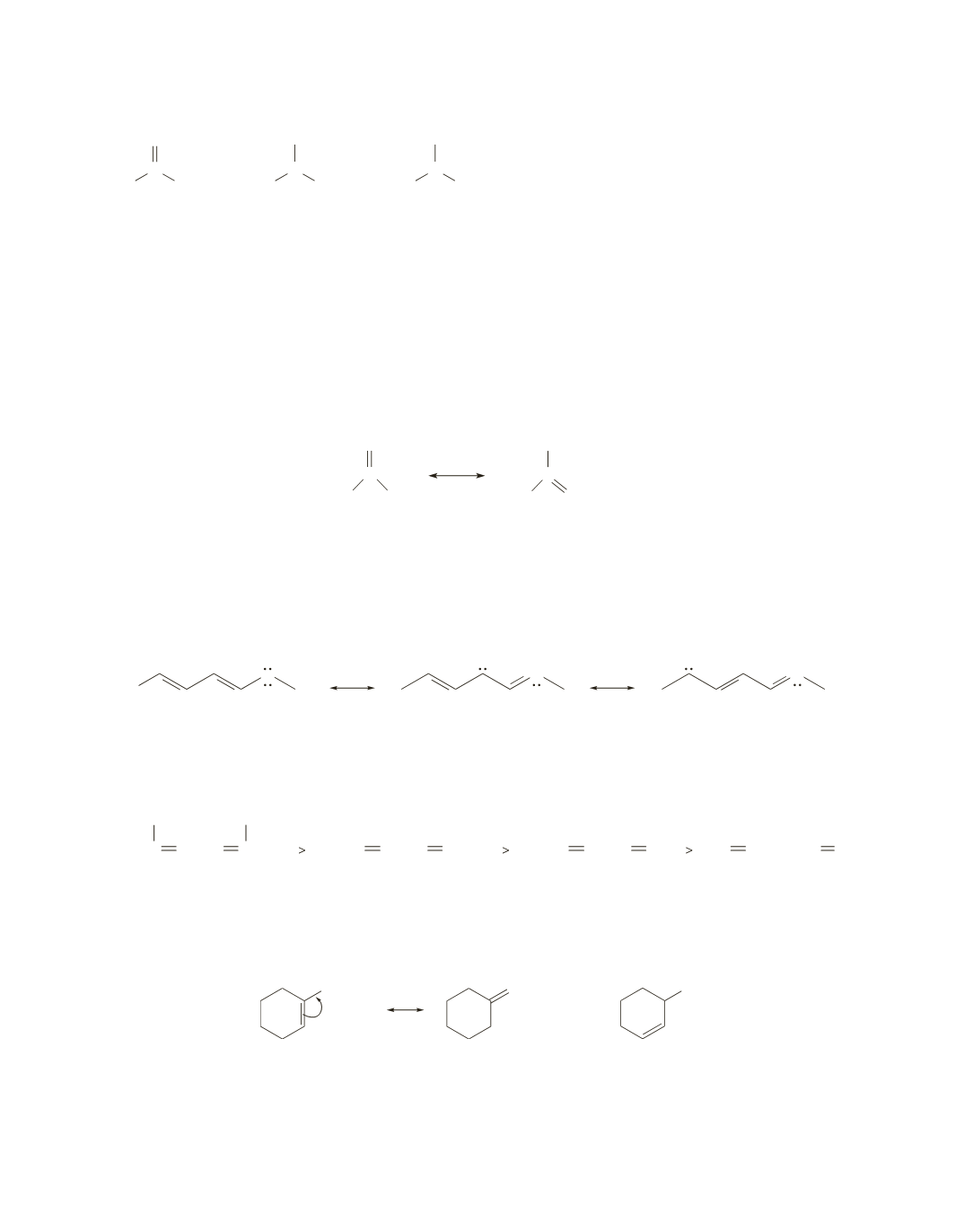
>
>
The dianion has the greatest delocalization energy; it has three relatively stable resonance contributors.
(See the answer to Problem 7.)
The monoanion is the next most stable; it has two relatively stable resonance contributors.
The neutral species has the smallest delocalization energy; it has one relatively stable resonance
contributor and one that has separated charges.
9.
The carboxylate ion has the greater delocalization energy because it has two equivalent relatively stable
resonance contributors.
CH
3
C
O
O
CH
3
C
O
O
−
−
In contrast, the diene has three resonance contributors but only one is relatively stable (see the top of
page 330 in the text).
10.
The compound on the left has the greater delocalization energy because the lone-pair electrons on
oxygen are delocalized, whereas the lone-pair electrons on oxygen in the compound on the right are not
delocalized.
O
O
O
+
+
−
−
11.
The smaller the heat of hydrogenation (the positive value of
∆
H
°
), the more stable the compound.
Therefore, the relative stabilities of the dienes are
conjugated diene
7
isolated diene
7
cumulated diene
12.
CH
2
CHCH
2
CH
1,4-pentadiene
1,3-pentadiene
2,4-hexadiene
CH
3
CH
CH
3
CH
CHCH
3
CH
2
CHCH
CHCH
CH
2
2,5-dimethyl-2,4-hexadiene
CHCH
CH
3
C
CH
3
CH
3
CCH
3
13.
a.
The compound with delocalized electrons is more stable than the compound in which all the electrons
are localized.
CHCH
3
+
CHCH
3
+
CHCH
3
+
electrons are delocalized
electrons are localized
















