
264 Chapter 8
Copyright © 2017 Pearson Education, Inc.
Solutions to Problems
1.
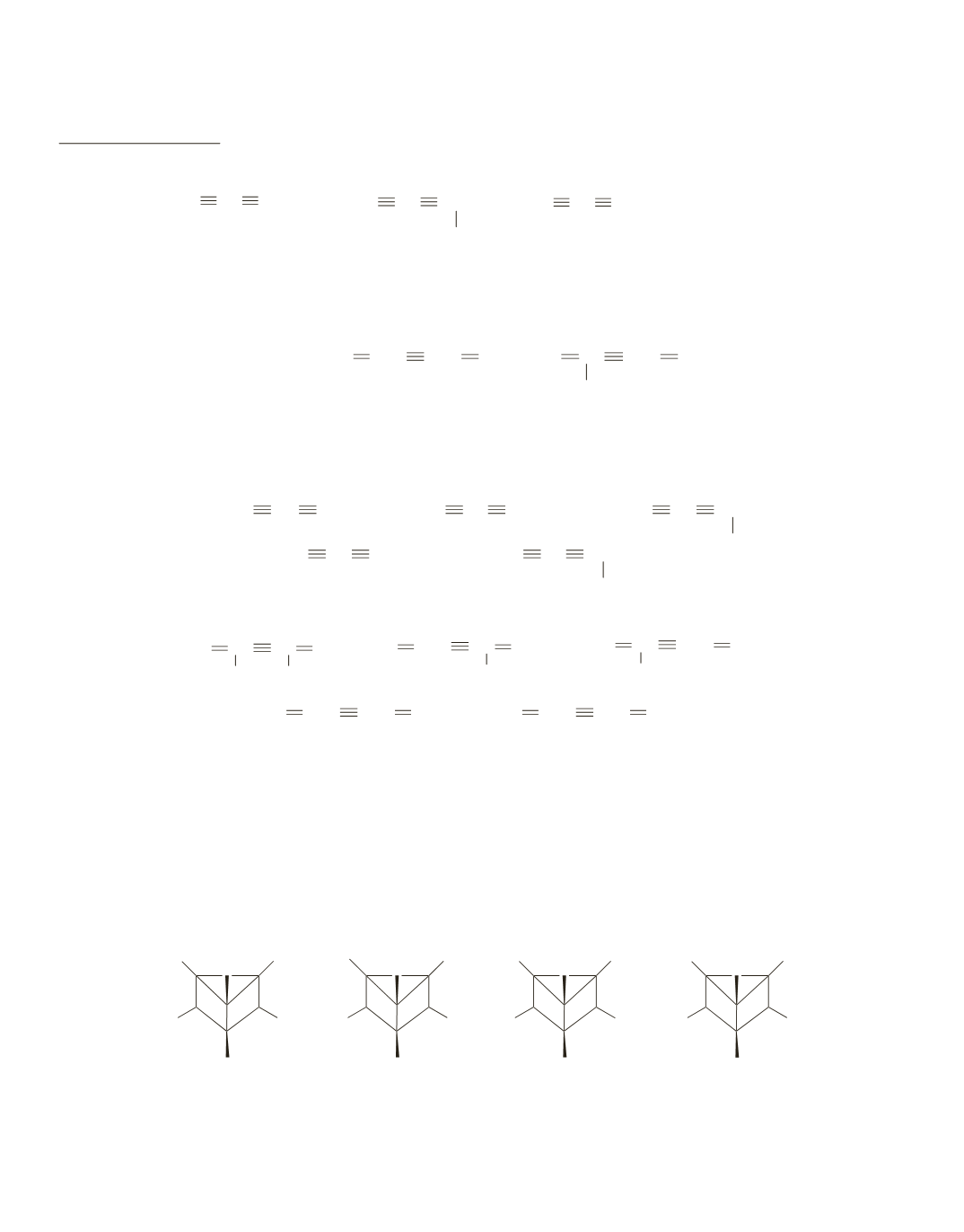
a. 1.
If stereoisomers are not included, three different monosubstituted compounds are possible.
BrC CC CCH
2
CH
3
HC CC CCHCH
3
Br
HC CC CCH
2
CH
2
Br
If stereoisomers are included, four different monosubstituted compounds are possible, because
the second listed compound has an asymmetric center so that it can have both the
R
and
S
configuration.
2.
If stereoisomers are not included, two different monosubstituted compounds are possible.
BrCH CHC CCH CH
2
CH
2
CC CCH
Br
CH
2
If stereoisomers are included, three different monosubstituted compounds are possible, because
the first listed compound has a double bond that can have cis-trans isomers.
b. 1.
If stereoisomers are not included, five different disubstituted compounds are possible.
HC CC CCH
2
CHBr
2
HC CC CCBr
2
CH
3
HC CC CCHCH
2
Br
Br
BrC CC CCH
2
CH
2
Br
BrC CC CCHCH
3
Br
2.
If stereoisomers are not included, five different disubstituted compounds are possible.
CH
2
CHC CCH CBr
2
CH
2
CHC CC CHBr
Br
CH
2
CC CCH CHBr
Br
CH
2
CC CC CH
2
Br
Br
BrCH CHC CCH CHBr
c. 1.
If stereoisomers are included, seven different disubstituted compounds are possible, because two
of the compounds have asymmetric centers so each can have either the
R
or
S
configuration.
2.
If stereoisomers are included, nine different disubstituted compounds are possible, because the
second and third compounds can have cis–trans isomers and the fifth compound can have cis–cis,
trans–trans, and cis–trans isomers. (Note that cis–trans is the same as trans–cis.)
2.
Ladenburg benzene is a better proposal. It would form one monosubstituted compound and three disubsti-
tuted compounds, in accordance with what early chemists knew about the structure of benzene.
H
H
Br
H
H
H
H
Br
Br
H
H
H
H
H
Br
H
Br
H
Br
H
Br
H
H
H
















